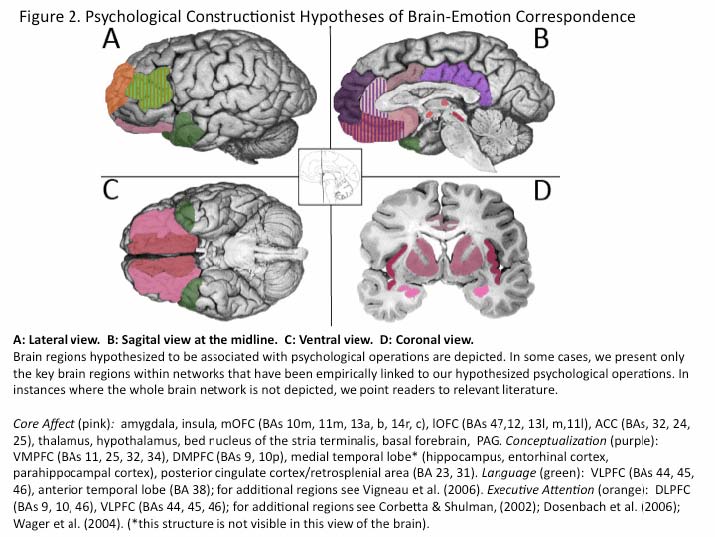
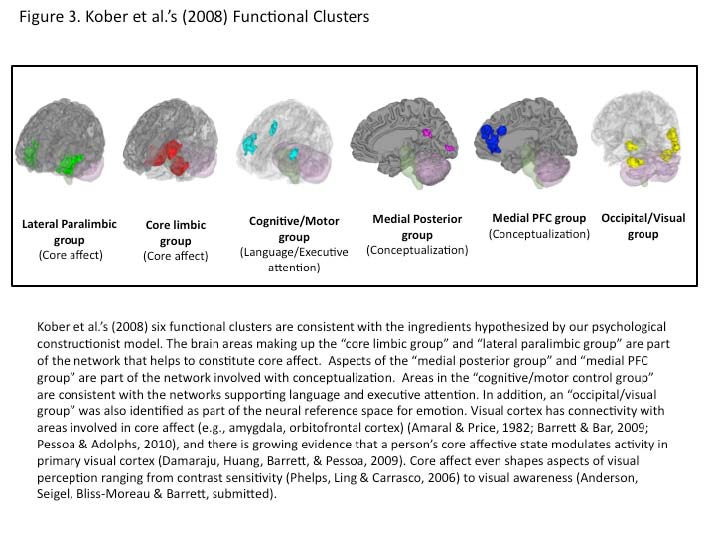
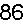Appendix
Aalto, S., Nänen, P., Wallius, E., Metsähonkala, L., Stenman, H., Niemi, P. M., et al. (2002). Neuroanatomical substrata of amusement and sadness: a PET activation study using film stimuli. Neuroreport 13: 67.
Ashwin, C., Baron-Cohen, S., Wheelwright, S., O’Riordan, M., & Bullmore, E. T. (2007). Differential activation of the amygdala and the ‘social brain’ during fearful face-processing in Asperger Syndrome. Neuropsychologia 45: 2–14.
Beauregard, M., Leroux, J. M., Bergman, S., Arzoumanian, Y., Beaudoin, G., Bourgouin, P., et al. (1998). The functional neuroanatomy of major depression: an fMRI study using an emotional activation paradigm. Neuroreport 9: 3253.
Blair, R. J. R., Morris, J. S., Frith, C. D., Perrett, D. I., & Dolan, R. J. (1999). Dissociable neural responses to facial expressions of sadness and anger. Brain 122: 883.
Breiter, H. C., Etcoff, N. L., Whalen, P. J., Kennedy, W. A., Rauch, S. L., Buckner, R. L., et al. (1996). Response and habituation of the human amygdala during visual processing of facial expression. Neuron 17: 875–888.
Britton, J. C., Phan, K. L., Taylor, S. F., Welsh, R. C., Berridge, K. C., & Liberzon, I. (2006). Neural correlates of social and nonsocial emotions: An fMRI study. Neuroimage 31: 397–409.
Britton, J. C., Taylor, S. F., Sudheimer, K. D., & Liberzon, I. (2006). Facial expressions and complex IAPS pictures: common and differential networks. Neuroimage 31: 906–919.
Buchanan, T. W., Lutz, K., Mirzazade, S., Specht, K., Shah, N. J., Zilles, K., et al. (2000). Recognition of emotional prosody and verbal components of spoken language: an fMRI study. Cognitive Brain Research 9: 227–238.
Calder, A. J., Beaver, J. D., Davis, M. H., van Ditzhuijzen, J., Keane, J., & Lawrence, A. D. (2007). Disgust sensitivity predicts the insula and pallidal response to pictures of disgusting foods. European Journal of Neuroscience 25: 3422–3428.
Cooney, R. E., Joormann, J., Atlas, L. Y., Eugène, F., & Gotlib, I. H. (2007). Remembering the good times: neural correlates of affect regulation. Neuroreport 18: 1771.
Damasio, A. R., Grabowski, T. J., Bechara, A., Damasio, H., Ponto, L. L., Parvizi, J., et al. (2000). Subcortical and cortical brain activity during the feeling of self-generated emotions. Nature Neuroscience 3: 1049–1056.
Dannlowski, U., Ohrmann, P., Bauer, J., Kugel, H., Arolt, V, Heindel, W., et al. (2007). Amygdala reactivity predicts automatic negative evaluations for facial emotions. Psychiatry Research: Neuroimaging 154: 13–20.
Das, P., Kemp, A. H., Liddell, B. J., Brown, K. J., Olivieri, G., Peduto, A., et al. (2005). Pathways for fear perception: modulation of amygdala activity by thalamo-cortical systems. Neuroimage 26: 141–148.
Deeley, Q., Daly, E. Surguladze, S., Tunstall, N., Mezey, G. Beer, D. et al. (2006). Facial emotion processing in criminal psychopathy: Preliminary functional magnetic resonance imaging study. The British Journal of Psychiatry 189: 533.
Dolan, R. J., Fletcher, P., Morris, J., Kapur, N., Deakin, J. F. W., & Frith, C. D. (1996). Neural activation during covert processing of positive emotional facial expressions. Neuroimage
4: 194–200.
Dougherty, D. D., Shin, L. M., Alpert, N. M., Pitman, R. K., Orr, S. P., Lasko, M., et al. (1999). Anger in healthy men: a PET study using script-driven imagery. Biological Psychiatry
46: 466–472.
Eugene, F., Lévesque, J., Mensour, B., Leroux, J. M., Beaudoin, G., Bourgouin, P., et al. (2003). The impact of individual differences on the neural circuitry underlying sadness. Neuroimage 19: 354–364.
Fischer, H., Fransson, P., Wright, C. I., & B\äckman, L. (2004). Enhanced occipital and anterior cingulate activation in men but not in women during exposure to angry and fearful male faces. Cognitive and Affective Behavioral Neuroscience 4: 326–334.
Fitzgerald, D. A., Angstadt, M., Jelsone, L. M., Nathan, P. J., & Phan, K. L. (2006). Beyond threat: amygdala reactivity across multiple expressions of facial affect. Neuroimage 30: 1441–1448.
Fitzgerald, D. A., Posse, S., Moore, G. J., Tancer, M. E., Nathan, P. J., & Phan, K. L. (2004). Neural correlates of internally-generated disgust via autobiographical recall: a functional magnetic resonance imaging investigation. Neuroscience letters 370: 91–96.
George, M. S., Parekh, P. I., Rosinsky, N., Ketter, T. A., Kimbrell, T. A., Heilman, K. M., et al. (1996). Understanding emotional prosody activates right hemisphere regions. Archives of Neurology 53: 665.
George, M. S., Ketter, T. A., Parekh, P. I., Horwitz, B., Herscovitch, P., & Post, R. M. (1995). Brain activity during transient sadness and happiness in healthy women. American Journal of Psychiatry 152: 341.
George, M. S., Ketter, T. A., Parekh, P. I., Rosinsky, N., Ring, H.,Casey, B. J., et al. (1994). Regional brain activity when selecting aresponse despite interference: an H2 15 O PET study of the Stroopand an emotional Stroop. Human Brain Mapping, 1, 194–209.
Grandjean, D., Sander, D., Pourtois, G., Schwartz, S., Seghier, M. L., Scherer, K. R., et al. (2005). The voices of wrath: brain responses to angry prosody in meaningless speech. Nature Neuroscience 8: 145–146.
Grezes, J., Pichon, S., & de Gelder, B. (2007). Perceiving fear in dynamic body expressions. Neuroimage 35: 959–967.
Grosbras, M. H., & Paus, T. (2006). Brain networks involved in viewing angry hands or faces. Cerebral Cortex 16: 1087.
Hutcherson, C. A., Goldin, P. R., Ochsner, K. N., Gabrieli, J. D., Barrett, L. F., & Gross, J. J. (2005). Attention and emotion: Does rating emotion alter neural responses to amusing and sad films? Neuroimage 27: 656–668.
Kesler/West, M. L., Andersen, A. H., Smith, C. D., Avison, M. J., Davis, C. E., Kryscio, R. J., et al. (2001). Neural substrates of facial emotion processing using fMRI. Cognitive Brain Research 11: 213–226.
Killgore, W. D., & Yurgelun-Todd, D. A. (2004). Activation of the amygdala and anterior cingulate during nonconscious processing of sad versus happy faces. Neuroimage 21: 1215–1223.
Kilts, C. D., Egan, G., Gideon, D. A., Ely, T. D., & Hoffman, J. M. (2003). Dissociable neural pathways are involved in the recognition of emotion in static and dynamic facial expressions. Neuroimage 18: 156–168.
Kimbrell, T. A., George, M. S., Parekh, P. I., Ketter, T. A., Podell, D. M., Danielson, A. L., et al. (1999). Regional brain activity during transient self-induced anxiety and anger in healthy adults. Biological Psychiatry 46: 454–465.
Lane, R. D., Reiman, E. M., Ahern, G. L., Schwartz, G. E., & Davidson, R. J. (1997). Neuroanatomical correlates of happiness, sadness, and disgust. American Journal of Psychiatry 154: 926–933.
Lange, K., Williams, L. M., Young, A. W., Bullmore, E. T., Brammer, M. J., Williams, S. C., et al. (2003). Task instructions modulate neural responses to fearful facial expressions. Biological Psychiatry 53: 226–232.
Lee, T. W., Josephs, O., Dolan, R. J., & Critchley, H. D. (2006). Imitating expressions: emotion-specific neural substrates in facial mimicry. Social cognitive and affective neuroscience
1: 122.
Lévesque, J., Eugene, F., Joanette, Y., Paquette, V., Mensour, B., Beaudoin, G., et al. (2003). Neural circuitry underlying voluntary suppression of sadness. Biological Psychiatry 53: 502–510.
Liddell, B. J., Brown, K. J., Kemp, A. H., Barton, M. J., Das, P., Peduto, A., et al. (2005). A direct brainstem–amygdala–cortical ‘alarm’system for subliminal signals of fear. Neuroimage 24: 235–243.
Liotti, M., Mayberg, H. S., Brannan, S. K., McGinnis, S., Jerabek, P., & Fox, P. T. (2000). Differential limbic–cortical correlates of sadness and anxiety in healthy subjects: implications for affective disorders. Biological Psychiatry 48: 30–42.
Malhi, G. S., Lagopoulos, J., Sachdev, P. S., Ivanovski, B., Shnier, R., & Ketter, T. (2007). Is a lack of disgust something to fear? A functional magnetic resonance imaging facial emotion recognition study in euthymic bipolar disorder patients. Bipolar disorders 9: 345.
Marci, C. D., Glick, D. M., Loh, R., & Dougherty, D. D. (2007). Autonomic and prefrontal cortex responses to autobiographical recall of emotions. Cognitive, Affective, & Behavioral Neuroscience 7: 243.
Mayberg, H. S., Liotti, M., Brannan, S. K., McGinnis, S., Mahurin, R. K., Jerabek, P. A., et al. (1999). Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. American Journal of Psychiatry 156: 675.
Minzenberg, M. J., Fan, J., New, A. S., Tang, C. Y., & Siever, L. J. (2007). Fronto-limbic dysfunction in response to facial emotion in borderline personality disorder: An event-related fMRI study. Psychiatry Research: Neuroimaging 155: 231–243.
Mitchell, D. G. V., Nakic, M., Fridberg, D., Kamel, N., Pine, D. S., & Blair, R. J. R. (2007). The impact of processing load on emotion. Neuroimage 34: 1299–1309.
Mitterschiffthaler, M. T., Fu, C. H., Dalton, J. A., Andrew, C. M., & Williams, S. C. (2007). A functional MRI study of happy and sad affective states induced by classical music. Human Brain Mapping 28: 1150–1162.
Mizuno, T., & Sugishita, M. (2007). Neural correlates underlying perception of tonality-related emotional contents. Neuroreport 18: 1651.
Moll, J., de Oliveira-Souza, R., Moll, F. T., Ignácio, F. A., Bramati, I. E., Caparelli-Dáquer, E. M., et al. (2005). The moral affiliations of disgust: a functional MRI study. Cognitive and Behavioral Neurology 18: 68.
Nomura, M., Ohira, H., Haneda, K., Iidaka, T., Sadato, N., Okada, T., et al. (2004). Functional association of the amygdala and ventral prefrontal cortex during cognitive evaluation of facial expressions primed by masked angry faces: an event-related fMRI study. Neuroimage 21: 352–363.
Ottowitz, W. E., Dougherty, D. D., Sirota, A., Niaura, R., Rauch, S. L., & Brown, W. A. (2004). Neural and endocrine correlates of sadness in women: implications for neural network regulation of HPA activity. Journal of Neuropsychiatry and Clinical Neurosciences 16:
446.
Paradiso, S., Robinson, R. G., Andreasen, N. C., Downhill, J. E., Davidson, R. J., Kirchner, P. T., et al. (1997). Emotional activation of limbic circuitry in elderly normal subjects in a PET study. American Journal of Psychiatry 154: 384.
Pardo, J. V., Pardo, P. J., & Raichle, M. E. (1993). Neural correlates of self-induced dysphoria. American Journal of Psychiatry 150: 713.
Partiot, A., Grafman, J., Sadato, N., Wachs, J., & Hallett, M. (1995). Brain activation during the generation of nonemotional and emotional plans. Neuroreport 6: 1397.
Peelen, M. V., Atkinson, A. P., Andersson, F., & Vuilleumier, P. (2007). Emotional modulation of body-selective visual areas. Social cognitive and affective neuroscience 2: 274.
Pessoa, L., McKenna, M., Gutierrez, E., & Ungerleider, L. G. (2002). Neural processing of emotional faces requires attention. Proceedings of the National Academy of Sciences 99: 11458.
Phillips, M. L., Bullmore, E. T., Howard, R., Woodruff, P. W., Wright, I. C., Williams, S. C., et al. (1998). Investigation of facial recognition memory and happy and sad facial expression perception: an fMRI study. Psychiatry Research: Neuroimaging 83: 127–138.
Phillips, M. L., Williams, L. M., Heining, M., Herba, C. M., Russell, T., Andrew, C., et al. (2004). Differential neural responses to overt and covert presentations of facial expressions of fear and disgust. Neuroimage 21: 1484–1496.
Phillips, M. L., Young, A. W., Scott, S. K., Calder, A. J., Andrew, C., Giampietro, V., et al. (1998). Neural responses to facial and vocal expressions of fear and disgust. Proceedings of the Royal Society B: Biological Sciences 265: 1809.
Phillips, M. L., Young, A. W., Senior, C., Brammer, M., Andrew, C., Calder, A. J., et al. (1997). A specific neural substrate for perceiving facial expressions of disgust. Nature 389: 495–
498.
Pietrini, P., Guazzelli, M., Basso, G., Jaffe, K., & Grafman, J. (2000). Neural correlates of imaginal aggressive behavior assessed by positron emission tomography in healthy subjects. American Journal of Psychiatry 157: 1772.
Pourtois, G., de Gelder, B., Bol, A., & Crommelinck, M. (2005). Perception of facial expressions and voices and of their combination in the human brain. Cortex 41: 49–60.
Rauch, A. V, Ohrmann, P., Bauer, J., Kugel, H., Engelien, A., Arolt, VS., et al. (2007). Cognitive coping style modulates neural responses to emotional faces in healthy humans: a 3-T FMRI study. Cerebral Cortex 17: 2526.
Reinders, A. A., den Boer, J. A., & Buchel, C. (2005). The robustness of perception. European Journal of Neuroscience 22: 524–530.
Salloum, J., Ramchandani, V., Bodurka, J., Rawlings, R., Momenan, R., George, D., et al. (2007). Blunted rostral anterior cingulate response during a simplified decoding task of negative emotional facial expressions in alcoholic patients. Alcoholism-Clinical and Experimental Research 31: 1490–1504.
Sambataro, F., Dimalta, S., Di Giorgio, A., Taurisano, P., Blasi, G., Scarabino, T., et al. (2006). Preferential responses in amygdala and insula during presentation of facial contempt and disgust. European Journal of Neuroscience 24: 2355–2362.
Sato, W., Yoshikawa, S., Kochiyama, T., & Matsumura, M. (2004). The amygdala processes the emotional significance of facial expressions: an fMRI investigation using the interaction between expression and face direction. Neuroimage 22: 1006–1013.
Schacher, M., Haemmerle, B., Woermann, F. G., Okujava, M., Huber, D., Grunwald, T., et al. (2006). Amygdala fMRI lateralizes temporal lobe epilepsy. Neurology 66: 81.
Schäfer, A., Schienle, A., & Vaitl, D. (2005). Stimulus type and design influence hemodynamic responses towards visual disgust and fear elicitors. International Journal of Psychophysiology 57: 53–59.
Schienle, A., Schäfer, A., Hermann, A., Walter, B., Stark, R., & Vaitl, D. (2006). fMRI responses to pictures of mutilation and contamination. Neuroscience letters 393: 174–
178. Schienle, A., Stark, R., Walter, B., Blecker, C., Ott, U., Kirsch, P., et al. (2002). The insula is not specifically involved in disgust processing: an fMRI study. Neuroreport 13: 2023.
Schroeder, U., Hennenlotter, A., Erhard, P., Haslinger, B., Stahl, R., Lange, K. W., et al. (2004). Functional neuroanatomy of perceiving surprised faces. Human brain mapping 23: 181–
187.
Silvert, L., Lepsien, J., Fragopanagos, N., Goolsby, B., Kiss, M., Taylor, J. G., et al. (2007). Influence of attentional demands on the processing of emotional facial expressions in the amygdala. NeuroImage 38: 357–366.
Simon, D., Craig, K. D., Miltner, W. H., & Rainville, P. (2006). Brain responses to dynamic facial expressions of pain. Pain 126: 309–318.
Somerville, L. H., Kim, H., Johnstone, T., Alexander, A. L., & Whalen, P. J. (2004). Human amygdala responses during presentation of happy and neutral faces: correlations with state anxiety. Biological Psychiatry 55: 897–903.
Sprengelmeyer, R., Rausch, M., Eysel, U. T., & Przuntek, H. (1998). Neural structures associated with recognition of facial expressions of basic emotions. Proceedings of the Royal Society B: Biological Sciences 265: 1927.
Stark, R., Schienle, A., Sarlo, M., Palomba, D., Walter, B., & Vaitl, D. (2005). Influences of disgust sensitivity on hemodynamic responses towards a disgust-inducing film clip. International Journal of Psychophysiology 57: 61–67.
Stark, R., Schienle, A., Walter, B., Kirsch, P., Sammer, G., Ott, U., et al. (2003). Hemodynamic responses to fear and disgust-inducing pictures: an fMRI study. International Journal of Psychophysiology 50: 225–234.
Stark, R., Zimmermann, M., Kagerer, S., Schienle, A., Walter, B., Weygandt, M., et al. (2007). Hemodynamic brain correlates of disgust and fear ratings. Neuroimage 37: 663–673.
Vuilleumier, P., Armony, J. L., Driver, J., & Dolan, R. J. (2001). Effects of attention and emotion on face processing in the human brain An event-related fMRI study. Neuron 30: 829–841.
Wang, L., McCarthy, G., Song, A. W., & Labar, K. S. (2005). Amygdala activation to sad pictures during high-field (4 tesla) functional magnetic resonance imaging. Emotion 5: 12–22.
Whalen, P. J., Shin, L. M., McInerney, S. C., Fischer, H., Wright, C. I., & Rauch, S. L. (2001). A functional MRI study of human amygdala responses to facial expressions of fear versus anger. Emotion1: 70–83.
Wicker, B., Keysers, C., Plailly, J., Royet, J. P., Gallese, V., & Rizzolatti, G. (2003). Both of us disgusted in My insula: the common neural basis of seeing and feeling disgust. Neuron
40: 655-664.
Williams, L. M., Brown, K. J., Das, P., Boucsein, W., Sokolov, E. N., Brammer, M. J., et al. (2004). The dynamics of cortico-amygdala and autonomic activity over the experimental
time course of fear perception. Cognitive Brain Research 21: 114–123.
Williams, L. M., Das, P., Liddell, B. J., Kemp, A. H., Rennie, C. J., & Gordon, E. (2006). Mode of functional connectivity in amygdala pathways dissociates level of awareness for signals of fear. Journal of Neuroscience 26:9264.
Williams, L. M., Kemp, A. H., Felmingham, K., Barton, M., Olivieri, G., Peduto, A., Gordon, E., & Bryant, R. A. (2006). Trauma modulates amygdala and medial prefrontal responses to consciously attended fear. Neuroimage 29: 347–357.
Williams, L. M., Liddell, B. J., et al. (2006). Amygdala-prefrontal dissociation of subliminal and supraliminal fear. Human brain mapping 27: 652–661.
Williams, L. M., Phillips, M. L., Brammer, M. J., Skerrett, D., Lagopoulos, J., Rennie, C., et al. (2001). Arousal dissociates amygdala and hippocampal fear responses: evidence from simultaneous fMRI and skin conductance recording. Neuroimage 14: 1070–1079.
Williams, M. A., McGlone, F., Abbott, D. F., & Mattingley, J. B. (2005). Differential amygdala responses to happy and fearful facial expressions depend on selective attention. Neuroimage 24: 417–425.
Wright, C. I., Wedig, M. M., Williams, D., Rauch, S. L., & Albert, M. S. (2006). Novel fearful faces activate the amygdala in healthy young and elderly adults. Neurobiology of Aging
27: 361–374. Wright, P., He, G., Shapira, N. A., Goodman, W. K., & Liu, Y. (2004). Disgust and the insula: fMRI responses to pictures of mutilation and contamination. Neuroreport 15: 2347.
Yamasaki, H., LaBar, K. S., & McCarthy, G. (2002). Dissociable prefrontal brain systems for attention and emotion. Proceedings of the National Academy of Sciences 99: 11447– 11451.
References
Addis, D. R., Wong, A. T. and Schacter, D. L. (2007). Remembering the past and imagining the future: Common and distinct neural substrates during event construction and elaboration. Neuropsychologia 45: 1363-1377. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed
Adolphs, R. (2008). Fear, faces, and the human amygdala. Current Opinion in Neurobiology 18:166-72. http://www.ncbi.nlm.nih.gov/pubmed/18655833 Adolphs, R. (2009). The social brain: Neural basis of social knowledge. Annual Review of Psychology 60: 693-716. http://www.ncbi.nlm.nih.gov/pubmed/18771388 Adolphs, R. (2010). What does the amygdala contribute to social cognition? Annals of the New York Academy of Sciences: The year in Cognitive Neuroscience: 1191:42-61.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2871162/
Adolphs, R., & Tranel, D. (1999). Intact recognition of emotional prosody following amygdala damage. Neuropsychologia 37: 1285-1292.
http://emotion.caltech.edu/papers/AdolphsTranel1999Intact.pdf
Adolphs, R., Gosselin, F., Buchanan, T. W., Tranel, D., Schyns, P., & Damasio, A. R. (2005). A mechanism for impaired fear recognition after amygdala damage. Nature 433: 68-72.
http://www.nature.com/nature/journal/v433/n7021/abs/nature03086.html
Adolphs, R., Tranel, D., Damasio, H., & Damasio, A. (1994). Impaired recognition of emotion in facial expressions following bilateral damage to the human amygdala. Nature 372: 669
672. http://www.nature.com/doifinder/10.1038/372669a0
Adolphs, R., Tranel, D., Damasio, H., & Damasio, A. R. (1995). Fear and the human amygdala. Journal of Neuroscience 15: 5879.
http://www.jneurosci.org/cgi/content/abstract/15/9/5879
Adolphs, R., Tranel, D., Hamann, S., Young, A. W., Calder, A. J., Phelps, E. A., et al. (1999). Recognition of facial emotion in nine individuals with bilateral amygdala damage. Neuropsychologia 37: 1111-1117.
http://www.cs.phs.uoa.gr/el/courses/emotions/papers/ADOPLHS%201999.pdf
Amaral, D. G., & Price, J. L. (1984). Amygdalo-cortical projections in the monkey (Macaca fascicularis). Journal of Comparative Neurology: 230 465-496.
http://www3.interscience.wiley.com/journal/109688079/abstract
Amodio, D.M., Master, S.L., Yee, C.M., Taylor, S. H. (2008). Neurocognitive components of the behavioral inhibition and activation systems. Implications for theories of self-regulation. Psychophysiology 45: 11-19.
An, X., Bandler, R., Ongur, D. & Price, J.L. (1998). Prefrontal cortical projections to longitudinal columns in the midbrain periaqueductal gray in macaque monkeys. The Journal of Comparative Neurology 401:455–479. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/9826273
Anderson, A.K. & Phelps, E.A. (1998). Intact recognition of vocal expressions of fear following bilateral lesions of the amygdala. NeuroReport 9: 3607-3613
http://www.ncbi.nlm.nih.gov/pubmed/9858368
Anderson, A.K., & Phelps, E.A. (2001). Lesions of the human amygdala impair enhanced perception of emotionally salient events. Nature 411: 305-309.
http://www.psych.nyu.edu/phelpslab/abstracts/lesions_amygdala.pdf
Anderson, A.K. & Phelps, E.A. (2002). Is the human amygdala critical for the subjective experience of emotion? Evidence of intact dispositional affect in patients with amygdala lesions. Journal of Cognitive Neuroscience 14: 709-720. http://www.psych.nyu.edu/phelpslab/abstracts/anderson_phelps2002.pdf
Anderson, M.L. (2010). Neural reuse: A fundamental organizational principle of the brain. Behavioral and Brain Sciences 33, 245–313. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/20964882
Angyal, A. (1941). Disgust and related aversions. Journal of Abnormal and Social Psychology
36: 393-412.
Aron, A.R., Robbins, T.W., & Poldrack, R.A., (2004). Inhibition and the right inferior frontal cortex. Trends in Cognitive Sciences 8: 170-177. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/15050513
Atkinson, A. P., Heberlein, A. S., & Adolphs, R. (2007). Spared ability to recognise fear from static and moving whole-body cues following bilateral amygdala damage. Neuropsychologia 45: 2772–2782. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1988781/
Bach, D.R., Schachinger, H. Neuhoff, J.G., Esposito, F. Di Salle, F., Lehmann, C., Herdener, M., Scheffler, K. & Seifrit, E. (2008). Rising sound intensity: An intrinsic warning cue activating the amygdala. Cerebral Cortex 18: 15-150 http://www.ncbi.nlm.nih.gov/pubmed?term=Rising%20sound%20intensity%3A%20An %20intrinsic%20warning%20cue%20activating%20the%20amygdala
Bachorowski, J.A., & Owren, M.J. (1995). Vocal expression of emotion: Acoustic properties of speech are associated with emotional intensity and context. Psychological Science 6: 219–224. http://pss.sagepub.com/content/6/4/219.abstract
Badre, D., & Wagner, A. D. (2007). Left ventrolateral prefrontal cortex and the cognitive control of memory. Neuropsychologia 45: 2883-2901. http://www.stanford.edu/group/memorylab/
Bancaud, J., Brunet-Bourgin, F., Chauvel, P., & Halgren, E. (1994). Anatomical origin of deja vu and vivid 'memories' in human temporal lobe epilepsy. Brain 117: 71-90 http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/8149215
Banich, M.T. (2009). Executive function: The search for an integrated account. Current Directions in Psychological Science, 18(2), 89-94. http://psych.colorado.edu/~mbanich
Bar, M. (2009). The proactive brain: memory for predictions. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 364: 1235-1243. doi: 10.1098/rstb.2008.0310. http://rstb.royalsocietypublishing.org/content/364/1521/1235.abstract
Bar, M., Kassam, K. S., Ghuman, A. S., Boshyan, J., Schmid, A. M., Dale, A. M., et al. (2006). Top-down facilitation of visual recognition. Proceedings of the National Academy of Sciences 103: 449-454. http://www.pnas.org/content/103/2/449.full
Barbas, H. (1988). Anatomic organization of basoventral and mediodorsal visual recipient prefrontal regions in the rhesus monkey. The Journal of Comparative Neurology 276: 313-342. http://www3.interscience.wiley.com/journal/109690199/abstract?CRETRY=1&SRETRY
=0
Barbas, H., & Pandya, D. N. (1989). Architecture and intrinsic connections of the prefrontal cortex in the rhesus monkey. The Journal of Comparative Neurology 286: 353-375.
http://www3.interscience.wiley.com/journal/109690672/abstract
Barbas, H., Henion, T. H., & Dermon, C. R. (1991). Diverse thalamic projections to the prefrontal cortex in the rhesus monkey. The Journal of Comparative Neurology 313: 65
94. http://www3.interscience.wiley.com/journal/109692035/abstract Barret, L. F., & Kensinger, E. (2010). Context is routinely encoded during emotion perception.
Psychological Science. Barrett, L. F. (2006a). Are emotions natural kinds? Perspectives on Psychological Science 1: 28.
http://www.bc.edu/sites/asi/publications/lfb/Barrett2006kinds.pdf
Barrett, L. F. (2006b). Solving the emotion paradox: Categorization and the experience of emotion. Personality and Social Psychology Review, 10: 20.
http://psr.sagepub.com/cgi/content/abstract/10/1/20
Barrett, L. F. (2009a). The future of psychology: Connecting mind to brain. Perspectives on Psychological Science 4: 326-339. http://www.affectivescience.org/pubs/2009/barrett2009-future-psych.pdf
Barrett, L. F. (2009b). Variety is the spice of life: A psychological construction approach to understanding variability in emotion. Cognition & Emotion, 23: 1284–1306.
http://www.affective-science.org/pubs/2009/variety-2009.pdf
Barrett, L. F., & Bar, M. (2009). See it with feeling: Affective predictions in the human brain. Philosophical Transactions of the Royal Society of London: Series B. Biological Sciences, 364: 1325–1334. http://barlab.mgh.harvard.edu/papers/RSBarrettBar.pdf
Barrett, L. F., & Bliss-Moreau, E. (2009). Affect as a psychological primitive. Advances in Experimental Social Psychology 41: 167–218.
http://www3.interscience.wiley.com/journal/122501435/abstract
Barrett, L. F., & Wager, T. D. (2006). The structure of emotion: Evidence from neuroimaging studies. Current Directions in Psychological Science 15: 79–83.
http://psycnet.apa.org/index.cfm?fa=search.displayRecord&uid=2006-06699-007
Barrett, L. F., Lindquist, K. A., & Gendron, M. (2007). Language as context for the perception of emotion. Trends in Cognitive Sciences 11: 327-332.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2225544/
Barrett, L. F., Lindquist, K. A., Bliss-Moreau, E., Duncan, S., Gendron, M., Mize, J., et al. (2007). Of mice and men: Natural kinds of emotions in the mammalian brain? A response to Panksepp and Izard. Perspectives on Psychological Science 2: 297.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2597798/
Barrett, L. F., Mesquita, B., Ochsner, K. N., & Gross, J. J. (2007). The experience of emotion. Annual Review of Psychology 58: 373-403.
http://arjournals.annualreviews.org/doi/full/10.1146/annurev.psych.58.110405.085709
Barrett, L. F., Ochsner, K. N., & Gross, J. J. (2007). On the automaticity of emotion. In: Ed. J. Bargh. Social psychology and the unconscious: The automaticity of higher mental processes. New York Psychology Press.
Barrett, L. F., Tugade, M. M., & Engle, R. W. (2004). Individual differences in working memory capacity and dual-process theories of the mind. Psychological Bulletin 130: 553–573.
http://psycnet.apa.org/index.cfm?fa=fulltext.journal&jcode=bul&vol=130&issue=4&for mat=html&page=553&expand=1
Barrett, L.F. (in press). Bridging token identity theory and supervenience theory through psychological construction. Psychological Inquiry.
Barsalou, L.W., & Wiemer-Hastings, K. (2005). Situating abstract concepts. In D. Pecher and R. Zwaan (Eds.), Grounding cognition: The role of perception and action in memory, language, and thought (pp. 129-163). New York: Cambridge University Press.
http://psychology.emory.edu/cognition/barsalou/papers/Barsalou_WiemerHastings_chap_2005_abstract_concepts.pdf
Barsalou, L.W. (2010). Grounded cognition: Past, present and future. Topics in Cognitive Science 2: 716-724.
http://psychology.emory.edu/cognition/barsalou/papers/Barsalou_topiCS_2010_grounded _cognition.pdf
Basole, A., White, L.E., & Fitzpatrick, D. (2003). Mapping multiple features in the population response of visual cortex. Nature 423: 986–990.
http://www.nature.com/nature/journal/v423/n6943/abs/nature01721.html
Bechara, A., Tranel, D., & Damasio, H. (2000). Characterization of the decision-making deficit of patients with ventromedial prefrontal cortex lesions. Brain: A Journal of Neurology
123: 2189-2202. http://brain.oxfordjournals.org/cgi/content/abstract/123/11/2189
Bechara, A., Tranel, D., Damasio, H., & Damasio, A. R. (1996). Failure to respond autonomically to anticipated future outcomes following damage to prefrontal cortex. Cerebral Cortex 6: 215-225. http://cercor.oxfordjournals.org/cgi/content/abstract/6/2/215
Bechara, A., Tranel, D., Damasio, H., Adolphs, R., Rockland, C., & Damasio, A. (1995). Double dissociation of conditioning and declarative knowledge relative to the amygdala and hippocampus in humans. Science 269: 1115-1118.
http://www.sciencemag.org/cgi/content/abstract/sci;269/5227/1115
Bechtel, W. (2008). Mental mechanisms. Philosophical perspectives on cognitive neuroscience. New York: Taylor and Francis Group. http://www.amazon.com/Mental-MechanismsPhilosophical-PerspectivesNeuroscience/dp/0805863346/ref=sr_1_3?s=books&ie=UTF8&qid=1296488925&sr=1-3
Beer, J.S., John, O., Scabini, D., Knight, R.T. (2003). Orbitofrontal cortex and social behavior. Integrating self-monitoring and emotion-cognition interactions. Journal of Cognitive Neuroscience 18: 871-879. http://www.ncbi.nlm.nih.gov/pubmed/14561114
Behbehani, M.M. (1995). Functional characteristics of the midbrain periaqueductal gray. Progress in Neurobiology 46, 575–605. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/8545545
Berridge, K. C., & Kringelbach, M. L. (2008). Affective neuroscience of pleasure: reward in humans and animals. Psychopharmacology 199: 457-480.
http://www.springerlink.com/content/h10384324x566083/
Blanchard, D.C., & Blanchard, R.J. (2003). What can animal aggression tell us about human aggression? Hormones and Behavior 44: 171-177.
http://www.ncbi.nlm.nih.gov/pubmed/14609539
Blackford, J.U., Buckholtz, J.W., Avery, S.N., Zald, D.H. (2010). A unique role for human amygdala in novelty detection. Neuroimage 50: 1188-1193.
http://www.ncbi.nlm.nih.gov/pubmed/20045069
Blakeslee, P. (1979). Attention and vigilance: performance and skin conductance response changes. Psychophysiology 16: 413-419.
http://www3.interscience.wiley.com/journal/119602640/abstract
Bliss-Moreau, E., Toscano, J.E., Baumann, M.D., Mason, W.A., & Amaral, D.G. (2010). Neonatal amygdala or hippocampus lesions influence responsiveness to objects. Developmental Psychobiology 52: 487-503. http://onlinelibrary.wiley.com/doi/10.1002/dev.20451/abstract
Blumberg, M. S., & Sokoloff, G. (2001). Do infant rats cry? Psychological Review 108: 83–95.
http://psycnet.apa.org/journals/rev/108/1/83.html
Bradley, M.M., Codispoti, M., Cuthbert, B.M., Lang, P.J. (2001). Emotion and Motivation I: Defensive and appetitive reactions in picture processing. Emotion 1: 276-296.
Brierley, B., Medford, N., Shaw, P., & David, A. S. (2004). Emotional memory and perception in temporal lobectomy patients with amygdala damage. British Medical Journal 75: 593. http://jnnp.bmj.com/content/75/4/593.abstract
Breiter, H.C., Etcoff, N.L., Whalen, P.J., Kennedy, W.A., Rauch, S.L., Buckner, R.L. et al. (1996). Response and habituation of the human amygdala. Neuron 17: 875-877. http://www.ncbi.nlm.nih.gov/pubmed/8938120
Büchel, C. Dolan, R.J., Armony, J.L., Friston, K.J. (1999). Amygdala-hippocampal involvement in human aversive trace conditioning revealed through event-related functional magnetic resonance imaging. Journal of Neuroscience 19: 10869-10876. http://www.ncbi.nlm.nih.gov/pubmed/10594068
Buckner, R. L., & Carroll, D. C. (2007). Self-projection and the brain. Trends in Cognitive Sciences 11: 49-57. http://www.ncbi.nlm.nih.gov/pubmed/17188554
Buckner, R. L., Andrews-Hanna, J. R., & Schacter, D. L. (2008). The brain's default network: anatomy, function, and relevance to disease. Annals of the New York Academy of Sciences 1124: 1-38. http://www.wjh.harvard.edu/~dsweb/pdfs/08_03_RLB_JRA_DLS.pdf
Buckner, R.L. and Vincent, J.L. (2007) Unrest at rest: Default activity and spontaneous network correlations. NeuroImage 37: 1091-1096. http://www.nmr.mgh.harvard.edu/nexus/publications/publications.html
Burns, L. H., Annett, L., Kelley, A. E., Everitt, B. J., & Robbins, T. W. (1996). Effects of lesions to amygdala, ventral subiculum, medial prefrontal cortex, and nucleus accumbens on the reaction to novelty: implication for limbic-striatal interactions. Behavioral Neuroscience
110: 60-73. http://psycnet.apa.org/journals/bne/110/1/60.html
Butter, C., & Snyder, D. (1972). Alterations in aversive and aggressive behaviors following orbital frontal lesions in rhesus monkeys. Acta Neurobiolagiae Experimentalis 32: 525
565. http://psycnet.apa.org/index.cfm?fa=search.displayRecord&uid=1973-08597-001 Cabanac, M. (2002). What is emotion? Behavioural Processes 60: 69-83.
http://www.sciencedirect.com/science?_ob=MImg&_imagekey=B6T2J-46VBRF3-14&_cdi=4920&_user=521319&_pii=S0376635702000785&_orig=search&_coverDate=1 2%2F31%2F2002&_sk=999399997&view=c&wchp=dGLbVzzzSkWz&md5=27571ed60e155373c08f5bd706a91f55&ie=/sdarticle.pdf
Cacioppo, J., Berntson, C., Larsen, J., Poehlmann, K., & Ito, T. (2000). The psychophysiology of emotion. In: The Handbook of emotions. M. Lewis and J.M. Haviland (Eds.) (2nd ed., pp. 173– 191). New York: Guilford.
Cacioppo, J., Berntson, C., & Nusbaum, H.C. (2008). Neuroimaging as a new tool in the toolbox of psychological science. Current Directions in Psychological Science 17: 62-67.
http://psychology.uchicago.edu/people/faculty/cacioppo/jtcreprints/cbn08.pdf
Calder, A. J. (2003). Disgust discussed. Annals of Neurology 53: 427-428.
http://www.ncbi.nlm.nih.gov/pubmed/12666109
Calder, A. J., Keane, J., Manes, F., Antoun, N., & Young, A. W. (2000). Impaired recognition and experience of disgust following brain injury. Nature Neuroscience 3: 1077–1078. http://psycnet.apa.org/index.cfm?fa=search.displayRecord&uid=2000-16375-003
Calder, A. J., Lawrence, A. D., & Young, A. W. (2001). Neuropsychology of fear and loathing. Nature Reviews Neuroscience 2: 352–363. http://jppsg.ac.uk/psych/resources/lawrence2001.pdf
Calder, A.J., Keane, J., Young, A.W., Lawrence, A.D, Mason, S. & Barker, R.A. (2010). The relation between anger and different forms of disgust. Implications for emotion recognition impairments in Huntington’s disease. Neuropsychologia 48: 2719-2729. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6T0D-50393XS2&_user=10&_coverDate=07%2F31%2F2010&_rdoc=1&_fmt=high&_orig=search&_or igin=search&_sort=d&_docanchor=&view=c&_searchStrId=1509825785&_rerunOrigin =scholar.google&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5=6 5288978413c0b945ef3d7058fd509cb&searchtype=a
Carmichael, S.T., Price, J.L. (1995). Sensory and premotor connections of the orbital and medial prefrontal cortex of macaque monkeys. Journal of Comparative Neurology 363:642-664. http://www.ncbi.nlm.nih.gov/pubmed/8847422
Carrive P, Bandler R, Dampney RA. (1989). Vicerotopic control of regional vascular beds by discrete groups of neurons within the midrain periaqueductal gray. Brain Research 493:385-90. http://www.ncbi.nlm.nih.gov/pubmed/2765906
Carver, C. S., & Harmon-Jones, E. (2009). Anger is an approach-related affect: evidence and implications. Psychological Bulletin 135: 183-204. http://psycnet.apa.org/index.cfm?fa=fulltext.journal&jcode=bul&vol=135&issue=2&for mat=html&page=183&expand=1
Champod, A.S. & Petrides, M. (2007). Dissociable roles of the posterior parietal and the prefrontal cortex in manipulation and monitoring processes. Proceedings of the National Academy of Sciences 104: 14837–14842. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/17804811
Chudasama, Y. & Robbins, T.W. (2003) Dissociable contributions of the orbitofrontal and infralimbic cortex to pavlovian autoshaping and discrimination reversal learning: Further evidence for the functional heterogeneity of the rodent frontal cortex. The Journal of Neuroscience 23: 8771-8780. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/14507977
Cheng, D. T., Richards, J., & Helmstetter, F. J. (2007). Activity in the human amygdala corresponds to early, rather than late period autonomic responses to a signal for shock. Learning & Memory 14: 485-490. http://learnmem.cshlp.org/content/14/7/485.abstract
Critchley, H. D., Mathias, C. J., Dolan, R.J. (2001). Neuroanatomical basis for first and second order representations of bodily states. Nature Neuroscience 4: 207-212.
http://www.ncbi.nlm.nih.gov/pubmed/11175883
Cole, M.W., & Schneider, W. (2007). The cognitive control network: Integrated cortical regions with dissociable functions. Neuroimage 37: 343-60.
http://www.ncbi.nlm.nih.gov/pubmed/17553704
Constantinidis, C., Williams, C.V., Goldman-Rakic, P.S. (2002) A role for inhibition in shaping the temporal flow of information in prefrontal cortex. Nature Neuroscience 5:175-180.
http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/11802172
Corbetta, M., & Shulman, G. L. (2002). Control of goal-directed and stimulus-driven attention in the brain. Nature Reviews Neuroscience 3: 215–229.
http://www.nature.com/nrn/journal/v3/n3/execsumm/nrn755.html
Corbetta, M., Patel, G., & Shulman, G. L. (2008). The reorienting system of the human brain: From environment to theory of mind. Neuron 58: 306–324.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2441869/
Costafreda, S.G., Brammer, M.J., David, A.S. & Fu, C.H.Y. (2008). Predictors of amygdala activation during the processing of emotional stimuli: A meta-analysis of 385 PET and fMRI studies. Brain Research Reviews 58: 57-50.
http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6SYS-4R40SJ01&_user=10&_coverDate=06%2F30%2F2008&_rdoc=1&_fmt=high&_orig=search&_or igin=search&_sort=d&_docanchor=&view=c&_acct=C000050221&_version=1&_urlVe rsion=0&_userid=10&md5=c0fb6a4930655607e64a98013f00d0fc&searchtype=a
Coyne, J. C. (1994). Self-reported distress: Analog or ersatz depression? Psychological Bulletin,
116: 29–29. http://psycnet.apa.org/journals/bul/116/1/29.html
Craig, A. D. (2002). How do you feel? Interoception: the sense of the physiological condition of the body. Nature Reviews Neuroscience 3: 655-666.
http://www.nature.com/nrn/journal/v3/n8/box/nrn894_BX1.html
Craig, A. D. B. (2009). How do you feel--now? The anterior insula and human awareness. Nature Reviews. Neuroscience 10: 59-70.
http://www.nature.com/nrn/journal/v10/n1/full/nrn2555.html
Cunningham, W. A., Van Bavel, J.J., Johnsen, I.R. (2008). Affective flexibility. Evaluative processing goals shape amygdala activity. Psychological Science, 19, 152-160.
http://faculty.psy.ohio-state.edu/cunningham/pdf/2008_Cunningham_etal_PS.pdf
Curtis, V., Aunger, R., & Rabie, T. (2004). Evidence that disgust evolved to protect from risk of disease. Proceedings of the Royal Society of London, Series B: Biological Sciences 271: 131–133. http://rspb.royalsocietypublishing.org/content/271/Suppl_4/S131.full.pdf
Damasio, A. (1999). The feeling of what happens: Body and emotion in the making of consciousness. New York: Random House. Damasio, A., Tranel, D., & Damasio, H. (1990). Individuals with sociopathic behavior caused by frontal damage fail to respond autonomically to social stimuli. Behavioral Brain Research
41: 81-94. http://www.ncbi.nlm.nih.gov/pubmed/2288668
Damsa, C., Kosel, M., & Moussally, J. (2009). Current status of brain imaging in anxiety disorders. Current Opinion in Psychiatry 22: 96-110. http://journals.lww.com/co
psychiatry/Abstract/2009/01000/Current_status_of_brain_imaging_in_anxiety.17.aspx
Davis, M. (1992). The role of the amygdala in fear and anxiety. Annual Review of Neuroscience
15: 353–375.
http://arjournals.annualreviews.org/doi/abs/10.1146/annurev.ne.15.030192.002033
Davis, M. Antoniadis, E., Amaral, D.G., & Winslow, J.T. (2008). Acoustic startle reflex in monkeys: A review. Reviews in the Neurosciences 19: 171-185.
http://www.ncbi.nlm.nih.gov/pubmed/18751523
de Bruin, J. P., Van Oyen, H. G., & Van De Poll, N. (1983). Behavioural changes following lesions of the orbital prefrontal cortex in male rats. Behavioural Brain Research 10: 209–
232. http://www.ncbi.nlm.nih.gov/pubmed/6686460
de Bruin, J.P. (1990). Social behavior in the prefrontal cortex. Progress in Brain Research 85: 495-496. http://www.ncbi.nlm.nih.gov/pubmed/2094911
Deco, G., Jirsa, V.K., McIntosh, A.R., (2011). Emerging concepts for the dynamical organization of resting-state activity in the brain. Nature Reviews Neuroscience 12: 43-56.
http://www.ncbi.nlm.nih.gov/pubmed/21170073
Deets, A.C., Harlow, H.F., Singh, S.D., Blomquist, A.J. (1970). Effects of bilateral lesions of the frontal granular cortex on the social behavior of rhesus monkeys. Journal of Comparative Physiological Psychology 72: 452-461. http://www.ncbi.nlm.nih.gov/sites/entrez
Devinsky, O., Morrell, M. J., & Vogt, B. A. (1995). Contributions of anterior cingulate cortex to behaviour. Brain 118: 279. http://brain.oxfordjournals.org/cgi/content/abstract/118/1/279
Dosenbach, N.U., Visscher, K.M., Palmer, E.D., Miezin, F.M., Wenger, K.K., Kang, H.C., et al. (2006). A core system for the implementation of task sets. Neuron 50: 799-812.
http://www.ncbi.nlm.nih.gov/pubmed/16731517
Duffy, E. (1934). Emotion: An example of the need for reorientation in psychology. Psychological Review 41: 184–198. http://psycnet.apa.org/journals/rev/41/2/184/
Duffy, E. (1957). The psychological significance of the concept of “arousal” or “activation.” Psychological Review 64: 265-275. http://psycnet.apa.org/journals/rev/64/5/265/ Duncan, S., & Barrett, L. F. (2007). Affect is a form of cognition: A neurobiological analysis. Cognition & Emotion 21: 1184. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2396787/ Eblen, F. & Graybiel A.M. (1995). Highly restricted origin of prefrontal cortical inputs to striosomes in the macaque monkey. Journal of Neuroscience 15: 5999-6013.
http://www.ncbi.nlm.nih.gov/sites/entrez
Edelman, G. M. (1989). The remembered present: A biological theory of consciousness. MIT Press. http://www.amazon.com/Remembered-Present-Biological-TheoryConsciousness/dp/046506910X
Eckert, M.A., Menon, V., Walczak, A., Ahlstrom, J., Denslow, S., Horowitz, A., Dubno, J.R. (2009). At the heart of the ventral attention system: The right anterior insula. Human Brain Mapping 30: 2530–2541http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pmc/articles/PMC2712290/
Ekman, P. & Cordaro, D. (in press). What is meant by calling emotions basic. Emotion Review. Ekman, P. (1999). Basic emotions. In T. Dalgleish & M. J. Powers (Eds.), Handbook of emotion and cognition. New York: John Wiley and Sons LTD.
http://onlinelibrary.wiley.com/book/10.1002/0470013494
Eslinger, P. J., & Damasio, A. R. (1985). Severe disturbance of higher cognition after bilateral frontal lobe ablation: Patient EVR. Neurology 35: 1731.
http://www.neurology.org/cgi/content/abstract/35/12/1731?ijkey=ae100b7b8d72e5bfebd 495730a37f463e8beb641&keytype2=tf_ipsecsha
Etkin, A., & Wager, T. D. (2007). Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. The American Journal of Psychiatry 164: 1476-1488.
http://ajp.psychiatryonline.org/cgi/content/abstract/164/10/1476
Ewbank, M. P., Barnard, P. J., Croucher, C. J., Ramponi, C., & Calder, A. J. (2009). The amygdala response to images with impact. Social Cognitive and Affective Neuroscience
4: 127-133.
Fanselow, M. S., & Poulos, A. M. (2005). The neuroscience of mammalian associative learning. Annual Review of Psychology 56: 207-234.
http://arjournals.annualreviews.org/doi/abs/10.1146/annurev..psych.56.091103.070213?u rl_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%3Dncbi.nlm.nih.gov
Feinstein, J.S., Adolphs, R., Damasio, A., Tranel, D. (2011). The human amygdala and the induction and experience of fear. Current Biology 21: 34-38.
http://www.ncbi.nlm.nih.gov/pubmed/21167712
Fendt, M., & Fanselow, M. S. (1999). The neuroanatomical and neurochemical basis of conditioned fear. Neuroscience and Biobehavioral Reviews 23: 743–760.
http://psycnet.apa.org/index.cfm?fa=search.displayRecord&uid=1999-05780-008
Ferris, C.F., Stolberg, T., Kulkarni, P., Murugavel, M. Blanchard, R., Blanchard, D.C., et al. (2008). Imaging the neural circuitry and chemical control of aggressive motivation. BMC Neuroscience 9: 1-35. http://www.biomedcentral.com/1471-2202/9/111
Ferry, A., Hespos, S.J., & Waxman, S. (2010). Language facilitates category formation in 3month-old infants. Child Development 81: 472 479.
http://groups.psych.northwestern.edu/infantcognitionlab/FerryHesposWaxman2010.pdf
Fischer, H., Wright, C. I., Whalen, P. J., McInerney, S. C., Shin, L. M., & Rauch, S. L. (2003). Brain habituation during repeated exposure to fearful and neutral faces: a functional MRI study. Brain Research Bulletin 59: 387-392.
http://www.ncbi.nlm.nih.gov/pubmed/12507690
Fontaine, J.R.J., Scherer, K.R., Roesch, E.B., & Ellsworth, P.C. (2007). The world of emotions is not two-dimensional. Psychological Science 18: 1050-1057.
http://pss.sagepub.com/content/18/12/1050.short
Fountoulakis, K.N., Giannakopoulos, P., Kövari, E., & Bouras, C. (2008). Assessing the role of cingulate cortex in bipolar disorder: Neuropathological, structural and functional imaging data. Brain Research Reviews 59: 9-21.
Fox, N. A. (1991). If it's not left, it's right. Electroencephalograph asymmetry and the development of emotion. The American Psychologist 46: 863-872.
http://psycnet.apa.org/index.cfm?fa=buy.optionToBuy&id=1991-32856001&CFID=6336004&CFTOKEN=47588819
Freedman, D. J., Riesenhuber, M., Poggio, T. & Miller, E. K. (2001). Categorical representation of visual stimuli in the primate prefrontal cortex. Science 291: 312–316.
http://www.sciencemag.org/content/291/5502/312.short
Freedman, D. J., Riesenhuber, M., Poggio, T., & Miller, E. K. (2002). Visual categorization and
the primate prefrontal cortex: neurophysiology and behavior. Journal of Neurophysiology
88: 929. http://jn.physiology.org/cgi/content/abstract/88/2/929 Friston, L. (2010). The free energy principle: A unified brain theory? Nature Reviews Neuroscience 11: 127-138
Fugate, J. M. B., Gouzoules, H., & Barrett, L. F. (2010). Reading chimpanzee faces: A test of the structural and conceptual hypotheses: Emotion 10: 544-554. http://www.affectivescience.org/pubs/2010/Fugate_Gouzoules_Barrett_2010CP.pdf
Fusar-Poli, P., Placentino, A., Carletti, F., Landi, P., Allen, P., Surguladze, S., et al. (2009). Functional atlas of emotional faces processing: a voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. Journal of Psychiatry & Neuroscience
34: 418-432. http://www.cma.ca/multimedia/staticContent/HTML/N0/l2/jpn/vol34/issue-6/pdf/pg418.pdf
Fuster, J. M. (2006). The cognit: a network model of cortical representation. International Journal of Psychophysiology 60: 125-132.
http://www.ncbi.nlm.nih.gov/pubmed/16626831
Gendron, M., & Barrett, F., (2009). Reconstructing the past: A century of ideas about emotion in psychology. Emotion Review 1: 316. http://www2.bc.edu/~barretli/pubs/2009/gendronbarrett-2009.pdf
Gendron, M., Lindquist, K.A., Wager, T.D., Bliss-Moreau, E., Kober, H. & Barrett, L.F., (in prep). Seeing with feeling: A meta-analytic review of visual cortex activity in emotion.
Gitelman, D.R., Nobre, A.C., Sonty, S., Parrish, T.B., Mesulam, M.M. (2005). Language network specializations: an analysis with parallel task designs and functional magnetic resonance imaging. Neuroimage 26: 975-985. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/15893473
Glenn, A.L., Raine, A., Schug, R.A. (2009). The neural correlates of moral decision making in psychopathy. Molecular Psychiatry 14: 5-9.
http://www.nature.com/mp/journal/v14/n1/abs/mp2008104a.html
Gotlib, I. H., & Hamilton, J. P. (2008). Neuroimaging and depression: Current status and unresolved issues. Current Directions in Psychological Science 17: 159.
http://psycnet.apa.org/?fa=main.doiLanding&uid=2008-04591-019
Goyer, P. F., Konicki, P. E., & Schulz, S. C. (1994). Brain imaging in personality disorders. In: Ed. K. M. Silk. Biological and Neurobehavioral Studies of Borderline Personality Disorder (pp. 109–125).
Grafman, J., Schwab, K., Warden, D., Pridgen, A., Brown, H. R., & Salazar, A. M. (1996). Frontal lobe injuries and violence a report of the Vietnam Head Injury Study. Neurology
46: 1231–1238. http://www.ncbi.nlm.nih.gov/pubmed/8628458
Greene, J. D., Nystrom, L. E., Engell, A. D., Darley, J. M., & Cohen, J. D. (2004). The neural bases of cognitive conflict and control in moral judgment. Neuron 44: 389–400.
http://www.wjh.harvard.edu/~jgreene/GreeneWJH/Greene-etal-Neuron04.pdf
Gregg, T.R., & Siegel, A. (2001). Brain structures and neurotransmitters regulating aggression in cats: implications for human aggression. Progress in Neuropsychopharacology Biological Psychiatry 25: 91-140. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/11263761
Grezes, J. & Decety, J. (2001). Functional anatomy of execution, mental simulation, observation, and verb generation of actions: A meta-analysis. Human Brain Mapping 12: 1-19. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/11198101
Grinband, J., Savitsky, J., Wager, T. D., Teichert, T., Ferrera, V. P., & Hirsch, J. (2010). The dorsal medial frontal cortex is sensitive to time on task, not response conflict or error likelihood. Neuroimage.
Gross, J. & Barrett, L.F. (in press). Emotion generation and emotion regulation. One or two depends on your point of view. Emotion Review.
Halgren, E., Walter, R.D., Cherlow, D.G., & Crandall, P.H. (1978). Mental phenomena evoked by electrical stimulation of the human hippocampal formation and amygdala. Brain 101: 83–117. http://www.ncbi.nlm.nih.gov/pubmed/638728
Harenski, C. L., Kim, S. H., & Hamann, S. (2009). Neuroticism and psychopathy predict brain activation during moral and nonmoral emotion regulation. Cognitive, Affective, & Behavioral Neuroscience 9: 1. http://www.ncbi.nlm.nih.gov/pubmed/19246323
Harlow, H. F., & Stagner, R. (1932). Psychology of feelings and emotions. I. Theory of feelings. Psychological Review 39: 570–589. http://psycnet.apa.org/journals/rev/39/6/570.pdf
Harmon-Jones, E., & Allen, J. J. (1998). Anger and frontal brain activity: EEG asymmetry consistent with approach motivation despite negative affective valence. Journal of Personality and Social Psychology 74: 1310–1316. http://psycnet.apa.org/index.cfm?fa=fulltext.journal&jcode=psp&vol=74&issue=5&form at=html&page=1310&expand=1
Harmon-Jones, E., & Sigelman, J. (2001). State anger and prefrontal brain activity: Evidence that insult-related relative left-prefrontal activation is associated with experienced anger and aggression. Journal of Personality and Social Psychology 80: 797–803. http://psycnet.apa.org/journals/psp/80/5/797.html
Harrison, N.A., Gray, M. A., Giarnos, P.J., Critchley, H. G. (2010). The embodiment of emotional feelings in the brain. The Journal of Neuroscience 30: 12878-12884. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/20861391
Hassabis, D. & Maguire, E.A. (2009). The construction system of the brain. Philosophical Transactions of the Royal Society B 364: 1263-1271. http://rstb.royalsocietypublishing.org/content/364/1521/1263.full
Haxby, J. V., Gobbini, M. I., Furey, M. L., Ishai, A., Schouten, J. L., & Pietrini, P. (2001). Distributed and overlapping representations of faces and objects in ventral temporal cortex. Science 293: 2425. http://www.sciencemag.org/cgi/content/abstract/sci;293/5539/2425
Herry, C., Bach, D. R., Esposito, F., Di Salle, F., Perrig, W. J., Scheffler, K., et al. (2007). Processing of temporal unpredictability in human and animal amygdala. Journal of Neuroscience 27: 5958. http://www.ncbi.nlm.nih.gov/pubmed/17537966
Hitchcock, J. M., & Davis, M. (1986). Lesions of the amygdala, but not of the cerebellum or red nucleus, block conditioned fear as measured with the potentiated startle paradigm. Behavioral Neuroscience 100: 11–22. http://psycnet.apa.org/journals/bne/100/1/11.html
Hitchcock, J. M., & Davis, M. (1987). Fear-potentiated startle using an auditory conditioned stimulus: effect of lesions of the amygdala. Physiology & Behavior 39: 403–408. http://www.ncbi.nlm.nih.gov/pubmed/3575483
Holland, P. C., & Gallagher, M. (1999). Amygdala circuitry in attentional and representational processes. Trends in Cognitive Sciences 3: 65–73. http://www.ncbi.nlm.nih.gov/pubmed/10234229
Hornak, J., Bramham, J., Rolls, E. T., Morris, R. G., O'Doherty, J., Bullock, P. R., et al. (2003). Changes in emotion after circumscribed surgical lesions of the orbitofrontal and cingulate cortices. Brain: A Journal of Neurology 126: 1691-1712. http://www.ncbi.nlm.nih.gov/pubmed/12805109
Horwitz, B. (2005). Integrating neuroscientific data across spatiotemporal scales. Comptes Rendus Biologies 328: 109-118. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/15770997
Hunt, W. A. (1941). Recent developments in the field of emotion. Psychological Bulletin 38: 249–276. http://psycnet.apa.org/?&fa=main.doiLanding&doi=10.1037/h0054615
Izard, C.E. (in press). Forms and functions in emotions: Matters of emotion-cognition interactions. Emotion Review.
Jabbi, M., Bastiaansen, J., & Keysers, C. (2008). A common anterior insula representation of disgust observation, experience and imagination shows divergent functional connectivity pathways. Plosone 3: 1-8.
James, W. (1884). What is an emotion? Mind 9: 188–205. http://www.jstor.org/pss/2246769
James, W. (1890/1998). Principles of Psychology. Chicago, IL: University of Chicago Press.
Jenison, R.L., Rangel, A., Oya, H. Kawasaki, H., Howard, M.A. (2011). Value encoding in single neurons in the human amygdala during decision making. The Journal of Neuroscience 31: 331–338. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/21209219
Jürgens, U. (2009). The Neural Control of Vocalization in Mammals: A Review. Journal of Voice 23: 1-10. http://www.ncbi.nlm.nih.gov/pubmed/8653176
Kamback, M. (1973). The effects of orbital and dorsolateral frontal cortical ablations on ethanol self-selection and emotional behaviors in monkeys. Neuropsychologia 11: 331-335. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/4209334
Keltner, D. Ekman, P., Gonzaga, G., & Beer, J. (2003). Facial expressions of emotion. In Eds.
R.J. Davidson, K.R. Scherer, H.H. Goldsmith. Handbook of affective sciences (p. 415427). New York Oxford University Press. http://www.amazon.com/Handbook-AffectiveSciences-Science/dp/0195126017
Kennedy, D.P., & Adolphs, R. (2010). Impaired fixation to eyes following amygdala damage arises from abnormal bottom-up attention. Neuropsychologia 48: 3392-3398. http://www.ncbi.nlm.nih.gov/pubmed/20600184
Kensinger, E. A., & Corkin, S. (2004). Two routes to emotional memory: Distinct neural processes for valence and arousal. Proceedings of the National Academy of Sciences 101: 3310–3315. http://www2.bc.edu/elizabeth-kensinger/Kensinger_Corkin_PNAS04.pdf
Kim, J.J., Rison, R.A., Fanselow, M.S., 1993. Effects of amygdala, hippocampus, and periaqueductal gray lesions on short- and long-term contextual fear. Behavioral Neuroscience 107: 1093–1098. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/8136063
Kipps, C.M., Duggins, A.J., McCusker, E.A., & Calder, A.J. (2007). Disgust and happiness recognition correlate with anteroventral insula and amygdala volume respectively in preclinical Huntington’s disease. Journal of Cognitive Neuroscience 19: 1206-1217.
http://www.ncbi.nlm.nih.gov/pubmed/17583995
Knutson, B., Rick, S., Wimmer, G. E., Prelec, D., & Loewenstein, G. (2007). Neural predictors of purchases. Neuron 53: 147-156. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1876732/
Kober, H., Barrett, L. F., Joseph, J., Bliss-Moreau, E., Lindquist, K., & Wager, T. D. (2008). Functional grouping and cortical-subcortical interactions in emotion: a meta-analysis of neuroimaging studies. NeuroImage 42: 998-1031. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2752702/
Koenigs M, Young L, Adolphs R, Tranel D, Cushman F, Hauser M, Damasio A. (2007). Damage to the prefrontal cortex increases utilitarian moral judgments. Nature 446: 908
11. http://www.ncbi.nlm.nih.gov/pubmed/17377536
Kopchia, K. L., Altman, H. J., & Commissaris, R. L. (1992). Effects of lesions of the central nucleus of the amygdala on anxiety-like behaviors in the rat. Pharmacology, Biochemistry, and Behavior 43: 453-461. http://www.ncbi.nlm.nih.gov/pubmed/1438482
Krain, A.L., Wilson, A.M., Arbuckle, R., Castellanos, F.X., Milham, M.P. (2006). Distinct neural mechanisms of risk and ambiguity: a meta-analysis of decision-making. Neuroimage 32: 477-484. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/16632383
Kringelbach, M. L., & Rolls, E. T. (2004). The functional neuroanatomy of the human orbitofrontal cortex: Evidence from neuroimaging and neuropsychology. Progress in Neurobiology 72: 341-372. http://www.kringelbach.dk/papers/PN_Kringelbach2004.pdf
LaBar, K. S., Gatenby, J. C., Gore, J. C., LeDoux, J. E., & Phelps, E. A. (1998). Human amygdala activation during conditioned fear acquisition and extinction: a mixed-trial fMRI study. Neuron 20: 937–945. http://www.ncbi.nlm.nih.gov/pubmed/9620698
LaBar, K. S., LeDoux, J. E., Spencer, D. D., & Phelps, E. A. (1995). Impaired fear conditioning following unilateral temporal lobectomy in humans. Journal of Neuroscience 15: 6846. http://www.ncbi.nlm.nih.gov/pubmed/7472442
Laine, C. M., Spitler, K. M., Mosher, C. P., & Gothard, K. M. (2009). Behavioral triggers of skin conductance responses and their neural correlates in the primate amygdala. Journal of Neurophysiology 101: 1749-1754. http://www.ncbi.nlm.nih.gov/pubmed/19144740
Lambon Ralph, M. A., Pobric, G., & Jefferies, E. (2009). Conceptual knowledge is underpinned by the temporal pole bilaterally: convergent evidence from rTMS. Cerebral Cortex 19: 832-838. http://www.ncbi.nlm.nih.gov/pubmed/18678765
Lang, P. J., Davis, M., & Öhman, A. (2000). Fear and anxiety: animal models and human cognitive psychophysiology. Journal of Affective Disorders 61: 137-159. http://www.stanford.edu/~kateri/Becky/PDFs/Lang%202000.pdf
LeDoux, J. (2007). The amygdala. Current Biology 17: R868-874.
LeDoux, J. E., Sakaguchi, A., Iwata, J., & Reis, D. J. (1985). Auditory emotional memories: establishment by projections from the medial geniculate nucleus to the posterior neostriatum and/or dorsal amygdala. Annals of the New York Academy of Sciences 444: 463-464. http://www.ncbi.nlm.nih.gov/pubmed/3860099
LeDoux, J.E., Ciccetti, P., Xagoraris, A., Romanski, L.M. (1990). The lateral amygdaloid nucleus: sensory interface of the amygdala in fear conditioning. Journal of Neuroscience
10: 1062-1069. http://www.ncbi.nlm.nih.gov/pubmed/2329367
LeDoux, J.E., Sakaguchi, A. & Reis, D.J (1983). Strain differences in fear between spontaneously hypertensive and normotensive rats. Brain Research 277: 137-143.
http://www.ncbi.nlm.nih.gov/pubmed/6640286
Lindquist, K. A., & Barrett, L. F. (2008). Constructing emotion: The experience of fear as a conceptual act. Psychological Science 19: 898.
http://www2.bc.edu/~lindqukr/docs/Lindquist&Barrett2008.pdf
Lindquist, K. A., Barrett, L. F., Bliss-Moreau, E., & Russell, J. A. (2006). Language and the perception of emotion. Emotion 6: 125-138.
http://nmr.mgh.harvard.edu/~lindqukr/publications.html
Lovick, T.A., 1992. Inhibitory modulation of the cardiovascular defence response by the ventrolateral periaqueductal grey matter in rats. Experimental Brain Research 89: 133–
139. http://www.springerlink.com/content/h18327q02g311765/
Machado, C.J., Bachevalier, J. (2006). The impact of selective amygdala, orbital frontal cortex, or hippocampal formation lesions on established social relationships in rhesus monkeys (Macaca mulatta). Behavioral Neuroscience 120:761-786.
http://www.ncbi.nlm.nih.gov/pubmed/16893284
MacLean, P.D., & Newman, J.D., (1988). Role of midline frontolimbic cortex in production of the isolation call of squirrel monkeys. Brain Research 450: 111-123.
http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/3401705
Mandler, G. (1975). Mind and emotion. New York: Wiley. Mandler, G. (1990). William James and the construction of emotion. Psychological Science 1: 179-180. http://www.jstor.org/stable/40062633
Mansouri, F.A., Tanaka, K., Buckley, M.J. (2009). Conflict-induced behavioral adjustment: A clue to the executive functions of prefrontal cortex. Nature Reviews Neuroscience 10: 141-152. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/19153577
Maskati, A.A.A & Zbrozyna, A.W. (1989). Cardiovascular and motor components of the defense reaction elicited in rats by electrical and chemical stimulation in amygdala. Journal of the Autonomic Nervous System 28: 127-132. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/2625501
Mason, W. A., Capitanio, J. P., Machado, C. J., Mendoza, S. P., & Amaral, D. G. (2006). Amygdalectomy and responsiveness to novelty in rhesus monkeys (Macaca mulatta): generality and individual consistency of effects. Emotion 6: 73-81. doi: 10.1037/15283542.6.1.73.
http://psycnet.apa.org/index.cfm?fa=fulltext.journal&jcode=emo&vol=6&issue=1&form at=html&page=73&expand=1
Matynh, P.W., (1983). Connections of midbrain periaqueductal gray in the monkey. I. Ascending efferent projections. Journal of Neurophysiology 49: 567-581.
http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/6300350
Mauss, I. B., & Robinson, M. D. (2009). Measures of emotion: A review. Cognition & Emotion
23: 209. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2756702/
Mayberg, H. S., Lozano, A. M., Voon, V., McNeely, H. E., Seminowicz, D., Hamani, C., et al. (2005). Deep brain stimulation for treatment-resistant depression. Neuron 45:651–660.
http://www.ncbi.nlm.nih.gov/pubmed/15748841
McDermott, K.B., Szpunar, K.K., & Christ, S.E. (2009). Laboratory-based and autobiographical retrieval tasks differ substantially in their neural substrates. Neuropsychologia 47: 22902298. http://memory.wustl.edu/McDermott_Lab/Publications.html
McIntosh, A. R., Bookstein, F. L., Haxby, J. V., & Grady, C. L. (1996). Spatial pattern analysis of functional brain images using partial least squares. Neuroimage 3: 143–157.
http://www.ncbi.nlm.nih.gov/pubmed/9345485
McIntosh, A. R. (2004). Contexts and catalysts: A resolution of the location and integration of function in the brain. Neuroinformatics 2: 175-181 http://www.springerlink.com.ezpprod1.hul.harvard.edu/content/k12p31883927q237/
Mendes, W. B., Blascovich, J., Hunter, S. B., Lickel, B., & Jost, J. T. (2007). Threatened by the unexpected: Physiological responses during social interactions with expectancy-violating partners. Journal of Personality and Social Psychology 92: 698.
http://psycnet.apa.org/journals/psp/92/4/698.html
Mesulam, M.M. & Mufson, E.J. (1982). Insula of the old world monkey. 111: Efferent cortical output and comments on function. The Journal of Comparative Neurology 212: 38-52.
http://www3.interscience.wiley.com/journal/109687236/abstract?CRETRY=1&SRETRY =0
Milders, M., Crawford, J. R., Lamb, A., & Simpson, S. A. (2003). Differential deficits in expression recognition in gene-carriers and patients with Huntington's disease. Neuropsychologia 41: 1484-1492. http://www.ncbi.nlm.nih.gov/pubmed/12849766
Milham, M.P., Banich, M.T., Webb, A., Barad, V., Cohen, N.J., Wszalek, T., Kramer, A.F., (2001). The relative involvement of anterior cingulate and prefrontal cortex in attentional control depends on nature of conflict. Brain Research. Cognitive Brain Research 12: 467
473. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/11689307
Miller, E. K. (2000). The prefrontal cortex and cognitive control. Nature Reviews Neuroscience
1: 59-65. http://www.doc.ic.ac.uk/~xh1/Referece/Visual-search/neural-cognitivecontrl.pdf
Miller, E. K., & Cohen, J. D. (2001). An integrative theory of prefrontal cortex function. Annual Review of Neuroscience 24: 167-202.
http://arjournals.annualreviews.org/doi/abs/10.1146%2Fannurev.neuro.24.1.167
Miller, E. K., Freedman, D. J., & Wallis, J. D. (2002). The prefrontal cortex: categories, concepts and cognition. Philosophical Transactions: Biological Sciences 357: 1123–1136.
http://rstb.royalsocietypublishing.org/content/357/1424/1123.full.pdf
Missilin, R., & Ropartz, P. (1981). Effects of lateral amygdala lesions on the responses to novelty in mice. Behavioural Processes 6: 329-336.
Mitchell, I. J., Heims, H., Neville, E. A., & Rickards, H. (2005). Huntington's disease patients show impaired perception of disgust in the gustatory and olfactory modalities. Journal of Neuropsychiatry and Clinical Neurosciences 17: 119.
http://www.ncbi.nlm.nih.gov/pubmed/15746492
Mitchell, J.P. (2009). Inferences about other people’s minds. Philosophical Transactions of the Royal Society B 364: 1309-1316.
http://www.wjh.harvard.edu/~scanlab/papers/2009_predictions_PhilTrans.pdf
Mobbs, D., Petrovic, P., Marchant, J.L., Hassabis, D., Weiskopf, N., Seymour, B., et al., (2007). When fear is near: threat imminence elicits prefrontal periaqueductal gray shifts in humans. Science 317: 1079. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2648508/
Moriguchi, Y. Negreira, A., Weirerich, M., Dautoff, R., Dickerson, B.C., Wright, C.I., Barrett, L.
F. (2010). Differential hemodynamic response in affective circuitry with aging: novely, valence and arousal. Journal of Cognitive Neuroscience.
http://www.mitpressjournals.org.ezpprod1.hul.harvard.edu/doi/pdf/10.1162/jocn.2010.21527
Moulton, S.T., & Kosslyn, S. (2009). Imagining predictions: Mental imagery as mental emulation. Philosophical Transactions of the Royal Society B 364: 1273-1280. http://rstb.royalsocietypublishing.org/content/364/1521/1273.full
Moyer, K.E., Kinds of aggression and their physiological basis, Communications in Behavioral Biology, A2 (1968) 65-87.
Murphy, F. C., Nimmo-Smith, I., & Lawrence, A. D. (2003). Functional neuroanatomy of emotions: A meta-analysis. Cognitive, Affective & Behavioral Neuroscience 3: 207-233. http://www.ncbi.nlm.nih.gov/pubmed/14672157
Nachman, M., & Ashe, J. H. (1974). Effects of basolateral amygdala lesions on neophobia, learned taste aversions, and sodium appetite in rats. Journal of Comparative and Physiological Psychology 87: 622-643. http://psycnet.apa.org/journals/com/87/4/622.pdf
Nelson, J.K., Reuter-Lorenz, P.A., Sylvester, C.C., Jonides, J. & Smith, E.E. (2003). Dissociable neural mechanisms underlying response-based and familiarity-based conflict in working memory. Proceedings of the National Academy of Sciences 100: 11171–11175. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/12958206
Newman, S. W. (1999). The medial extended amygdala in male reproductive behavior. A node in the mammalian social behavior network. Annals of the New York Academy of Sciences 877: 242-57. http://www.ncbi.nlm.nih.gov/sites/entrez
Northoff, (2006). Is emotion regulation self-regulation? Trends in Cognitive Sciences 9: 408
409. http://dept.psych.columbia.edu/~kochsner/pdf/Northoff_Tics_comment.pdf
Numan, M. (2007). Motivational systems and the neural circuitry of maternal behavior in the rat. Developmental Psychobiology 49: 12-21. http://www.ncbi.nlm.nih.gov/pubmed/17186513
Ochsner, K. N., Ray, R. D., Cooper, J. C., Robertson, E. R., Chopra, S., Gabrieli, J. D. E., et al. (2004). For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion. NeuroImage 23:483-499. http://www.ncbi.nlm.nih.gov/pubmed/15488398
Ochsner, K. N., Knierim, K., Ludlow, D., Hanelin, J., Ramachandran, T. & Mackey, S. (2004). Reflecting upon feelings: An fMRI study of neural systems supporting the attribution of emotion to self and other. Journal of Cognitive Neuroscience 16: 1748-1772. http://dept.psych.columbia.edu/~kochsner/pdf/Ochsner_Reflect_Feelings.pdf
Öhman, A. (2009). Of snakes and faces: An evolutionary perspective on the psychology of fear. Scandinavian Journal of Psychology 50: 543-552. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/19930253
Öngür, D., & Price, J. L. (1998). Prefrontal cortical projections to the hypothalamus in macaque monkeys. Journal of Comparative Neurology 30: 480-505. http://www3.interscience.wiley.com/journal/5005316/abstract
Öngür, D., & Price, J. L. (2000). The organization of networks within the orbital and medial prefrontal cortex of rats, monkeys and humans. Cerebral Cortex 10: 206.
http://cercor.oxfordjournals.org/cgi/content/abstract/10/3/206
Ortigue, S., Grafton, S. T., & Bianchi-Demicheli, F. (2007). Correlation between insula activation and self-reported quality of orgasm in women. NeuroImage 37: 551-560. http://www.ncbi.nlm.nih.gov/pubmed/17601749
Ortony A, & Turner, T.J. (1990). What’s basic about basic emotions? Psychological Review 97:315–331.http://web.ebscohost.com.ezpprod1.hul.harvard.edu/ehost/detail?hid=10&sid=79dcf313-fa01-4d4c-aff0542b9341511a%40sessionmgr13&vid=4&bdata=JnNpdGU9ZWhvc3QtbGl2ZSZzY29w ZT1zaXRl#db=psyh&AN=1990-27526-001
Owen, A.M., McMillan, K., Laird, A.R., Bullmore, E. (2005). N-Back working memory paradigm: A meta-analysis of normative functional neuroimaging studies. Human Brain Mapping 25: 46 –59. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/15846822
Panksepp, J. & Watt, D. (in press). What is basic about basic emotions? Lasting lessons from affective neuroscience. Emotion Review.
Panksepp, J. (1998). Affective neuroscience: The foundations of human and animal emotions. New York: Oxford University Press.
Panksepp, J. (2007). Neurologizing the psychology of affects: How appraisal-based constructivism and basic emotion theory can coexist. Perspectives on Psychological Science 2: 281. http://www3.interscience.wiley.com/journal/118509152/abstract
Paton, J.P., Belova, M.A., Morrison, S.E. & Salzman, C.D. (2006). The primate amygdala represents the positive and negative value of visual stimuli during learning. Nature 16: 865-870. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2396495/
Paus, T. (2001). Primate anterior cingulate cortex: Where motor control, drive and cognition interface. Nature Reviews Neuroscience 2: 417–424. http://www.oise.utoronto.ca/research/brainwaves/phpwebsite/files/uplink/Paus_2001.pdf
Penfield, W., & Falk, M.E. (1955). The insula. Further observations on its function. Brain 78: 445-470. http://brain.oxfordjournals.org/content/78/4/445.extract
Pessoa, L. (2010). Emotion and cognition and the amygdala: From “what is it?” to “what's to be done?”. Neuropsychologia 48: 3416-3429. http://www.indiana.edu/%7Elceiub/publications_files/Pessoa_Neuropsychologia_2010.p df
Pessoa, L. & Adolphs, R. (2010). Emotion processing and the amygdala: from a ‘low road’ to ‘many roads’ of evaluating biological significance. Nature Reviews Neuroscience 11: 773-783. http://www.indiana.edu/%7Elceiub/publications_files/Pessoa_Adolphs_NRN_2010_with _suppl.pdf
Pessoa, L. (2008). On the relationship between emotion and cognition. Nature Reviews Neuroscience 9: 148-158.
Petrides, M. (2005). Lateral prefrontal cortex: architectonic and functional organization.
Philosophical Transactions of the Royla Society of London: B Biological Sciences 360:781-95. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/15937012
Petrides, M. (2007). The orbitofrontal cortex: novelty, deviation from expectation, and memory. Annals of the New York Academy of Sciences 1121: 33-53. http://www.ncbi.nlm.nih.gov/pubmed/17872393
Phan, K. L., Wager, T., Taylor, S. F., & Liberzon, I. (2002). Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in PET and fMRI. NeuroImage
16: 331-348. http://www.ncbi.nlm.nih.gov/pubmed/12030820
Pobric, G., Jefferies, E., & Ralph, M. A. L. (2007). Anterior temporal lobes mediate semantic representation: mimicking semantic dementia by using rTMS in normal participants. Proceedings of the National Academy of Science 104: 20137-20141. http://www.pnas.org/content/104/50/20137.abstract
Poldrack, R.A., Halchenko, Y.O., Hanson, S.J. (2009). Decoding the large-scale structure of brain function by classifying mental States across individuals. Psychological Science 20: 1364-1372. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/19883493
Price, C.J., & Friston, K.J. (2005). Functional ontologies for cognition: The systematic definition of structure and function. Cognitive Neuropsychology 22: 262-275. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/21038249
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G.
L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences 98: 676-682. http://www.pnas.org/content/98/2/676.long
Raine, A., Lencz, T., Bihrle, S., LaCasse, L., & Colletti, P. (2000). Reduced prefrontal gray matter volume and reduced autonomic activity in antisocial personality disorder. Archives of General Psychiatry 57: 119-127. http://www.ncbi.nlm.nih.gov/pubmed/10665614
Raleigh, M. J., Steklis, H. D., Ervin, F. R., Kling, A. S., & McGuire, M. T. (1979). The effects of orbitofrontal lesions on the aggressive behavior of vervet monkeys (Cercopithecus aethiops sabaeus). Experimental Neurology 66: 158-168. http://www.ncbi.nlm.nih.gov/pubmed/113236
Rempel-Clower, N.L., & Barbas H. (1998). Topographic organization of connections between the hypothalamus and prefrontal cortex in the rhesus monkey. Journal of Comparative Neurology 398: 393-419. http://www.ncbi.nlm.nih.gov/pubmed/9714151
Rankin, K.P. Gorno-Tempini, M.L., Allison, S.C., Stanley, C.M., Glenn, S., Weiner, M.W., Miller, B.L. (2006). Structural anatomy of empathy in neurodegenerative disease. Brain
129: 2945-2956.
Reynolds, S. M., & Berridge, K. C. (2002). Positive and negative motivation in nucleus accumbens shell: Bivalent rostrocaudal gradients for GABA-elicited eating, taste "liking"/"disliking" reactions, place preference/avoidance, and fear. The Journal of Neuroscience 22: 7308-7320. http://www.jneurosci.org/cgi/content/abstract/22/16/7308 Reynolds, S. M., & Berridge, K. C. (2003). Glutamate motivational ensembles in nucleus accumbens: Rostrocaudal shell gradients of fear and feeding. European Journal of Neuroscience 17: 2187–2200. http://www.ncbi.nlm.nih.gov/pubmed/12786986
Reynolds, S. M., & Berridge, K. C. (2008). Emotional environments retune the valence of appetitive versus fearful functions in nucleus accumbens. Nature Neuroscience 11: 423
425. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2717027/
Roberson, D., & Davidoff, J. (2000). The categorical perception of color and facial expressions: The effect of verbal interference. Memory & Cognition 28: 977–986. http://www.ncbi.nlm.nih.gov/pubmed/11105523
Roberson, D., Damjanovic, L. & Pilling, M. (2007).Categorical perception of facial expressions: Evidence for a 'category adjustment' model. Memory & Cognition 35: 1814-1829.
Rogers, T. T., Lambon Ralph, M. A., Garrard, P., Bozeat, S., McClelland, J. L., Hodges, J. R., et al. (2004). Structure and deterioration of semantic memory: A neuropsychological and computational investigation. Psychological Review 111: 205-235. http://charlotte.cnbc.cmu.edu/ibsc/papers/RogersETAL04_PR.pdf
Rolls E.T., Critchley, H.D., Mason, R., Wakeman EA. (1996). Orbitofrontal cortex neurons: role in olfactory and visual association learning. Journal of Neurophysiology 75: 1970-1981. http://www.ncbi.nlm.nih.gov/pubmed/8734596
Rolls, E. (1999). The function of the orbitofrontal cortex. Neurocase 5: 301-312.
http://www.oxcns.org/papers/357_Rolls04.pdf
Rolls, E.T., Hornak, J., Wade, D., & McGrath J. (1994). Emotion-related learning in patients with social and emotional changes associated with frontal lobe damage. Journal of Neurology, Neurosurgery and Psychiatry 57:1518-1524. http://www.ncbi.nlm.nih.gov/pubmed/7798983
Roseman, I.J. (1984). Cognitive determinants of emotion: A structural theory. Review of Personality & Social Psychology 5: 11-36. http://web.ebscohost.com.ezpprod1.hul.harvard.edu/ehost/results?hid=10&sid=79dcf313-fa01-4d4c-aff0542b9341511a%40sessionmgr13&vid=5&bquery=%28AU+%28Roseman%29%29+and +%28DT+1984%29&bdata=JmRiPXBzeWgmdHlwZT0xJnNpdGU9ZWhvc3QtbGl2ZS ZzY29wZT1zaXRl
Rosen, H. J., Pace-Savitsky, K., Perry, R. J., Kramer, J. H., Miller, B. L., & Levenson, R. W. (2004). Recognition of emotion in the frontal and temporal variants of frontotemporal dementia. Dementia and Geriatric Cognitive Disorders 17: 277-281. http://www.ncbi.nlm.nih.gov/pubmed/15178936
Rosen, J.B. & Davis, M. (1988). Enhancement of acoustic startle by electrical stimulation of the amygdala. Behavioral Neuroscience 102: 195-202. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6WY4-46X46NT4F&_user=10&_coverDate=04%2F30%2F1988&_rdoc=1&_fmt=high&_orig=search&_ origin=search&_sort=d&_docanchor=&view=c&_searchStrId=1509840975&_rerunOrigi n=scholar.google&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5= e681bf616267abecaf65243450732d0d&searchtype=a
Rozin, P., Haidt, J., & McCaule, C. (2008). Disgust. In: The Handbook of Emotions. Eds. M. Lewis, J.M. Haviland-Jones and L.F. Barrett. (3rd ed. pp. 757-776). New York: Guilford.
Rozin, P., Haidt, J., & McCauley, C. (2000). Disgust. In: Eds. M. Lewis and J.M. Havilland-Jones. Handbook of emotions (2nd ed., pp. 637-653). New York: Guilford.
Rushworth, M.F., Buckley, M.J., Behrens, T.E., Walton, M.E., Bannerman, D.M. (2007). Functional organization of the medial frontal cortex. Current Opinion in Neurobiology
17: 220-227. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/17350820
Russell, J. A. (2003). Core affect and the psychological construction of emotion. Psychological Review 110: 145–172.
http://psycnet.apa.org/index.cfm?fa=fulltext.journal&jcode=rev&vol=110&issue=1&for mat=html&page=145&expand=1
Russell, J.A., Bachorowski, J., & Fernandez-Dols, J.M. (2003). Facial and vocal expressions of emotion. Annual Review of Psychology 54: 329-349.
https://www2.bc.edu/~russeljm/publications/annurev.psych.2003.pdf
Russell, J. A., & Barrett, L. F. (1999). Core affect, prototypical emotional episodes, and other things called emotion: Dissecting the elephant. Journal of Personality and Social Psychology 76: 805-819. http://psycnet.apa.org/journals/psp/76/5/805.html
Russell, J.A., & Bullock, M. (1986). On the dimensions preschoolers use to interpret facial expressions of emotion. Developmental Psychology 22: 97-102.
http://www2.bc.edu/~russeljm/
Sander, D., Grafman, J., & Zalla, T. (2003). The human amygdala: An evolved system for relevance detection. Reviews in the Neurosciences 14: 303–316.
http://www.affective-sciences.org/system/files/Sander_etal_RevNeuro.PDF
Sander, D., Grandjean, D, Kaiser, S., Wehrle, T., & Scherer, K.R. (2007). Interaction effects of perceived gaze direction and dynamic facial expression: Evidence for appraisal theories of emotion. European Journal of Cognitive Psychology 19: 470-480. http://www.lemanicneuroscience.ch/PENSTrainingCenter/articles/dsdg_ejcp06.pdf
Saver, J. L., & Damasio, A. R. (1991). Preserved access and processing of social knowledge in a patient with acquired sociopathy due to ventromedial frontal damage. Neuropsychologia
29: 1241-1249. http://www.ncbi.nlm.nih.gov/pubmed/1791934
Saxe, R. & Kanwisher, N. (2003). People thinking about people: The role of the temporoparietal junction in “theory of mind.” Neuroimage 19: 1835-1842.
http://web.mit.edu/bcs/nklab/media/pdfs/SaxeKanwisherNeuroImage03.pdf
Schachter, S., & Singer, J. (1962). Cognitive, social, and physiological determinants of an emotional state. Psychological Review 69: 379-399.
http://www.ncbi.nlm.nih.gov/pubmed/14497895
Schacter, D. L., Addis, D. R., & Buckner, R. L. (2007). Remembering the past to imagine the future: the prospective brain. Nature Reviews Neuroscience 8: 657-661.
http://www.nature.com/nrn/journal/v8/n9/abs/nrn2213.html
Shackman, A.J., Salomons, T.V., Slagter, H.A., Fox, A.S., Winter, J.J., Davidson, R.J. (2011). The integration of negative affect, pain, and cognitive control in the cingulate cortex. Nature Reviews Neuroscience 12: 154-167. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/21331082
Scherer, K.R., & Ellsworth, P.C. (2003). Appraisal processes in emotion. In R.J. Davidson, K.R. Scherer, & H. Goldsmith (Eds.), Handbook of affective sciences (pp. 572–589). New York: Oxford University Press. http://www.amazon.com/Handbook-Affective-SciencesScience/dp/0195126017
Schienle, A., & Schäfer, A. (2009). In search of specificity: functional MRI in the study of emotional experience. International Journal of Psychophysiology 73: 22-26.
http://www.ncbi.nlm.nih.gov/pubmed/19530276
Schnur, T. T., Schwartz, M. F., Kimberg, D. Y., Hirshorn, E., Coslett, H. B., & Thompson-Schill, S. L. (2009). Localizing interference during naming: Convergent neuroimaging and neuropsychological evidence for the function of Broca's area. Proceedings of the National Academy of Sciences 106: 322-327. http://nihongo.j
talk.com/kanji/?link=http://www.pnas.org/content/106/1/322.full
Schwartz, C. E., Wright, C. I., Shin, L. M., Kagan, J., Whalen, P. J., McMullin, K. G., et al. (2003). Differential amygdalar response to novel versus newly familiar neutral faces: a functional MRI probe developed for studying inhibited temperament. Biological Psychiatry 53: 854-862. http://www.whalenlab.info/Publications%20Page/PDFs/Schwartz_2003.pdf
Shultz, W. (2006). Behavioral theories and the neurophysiology of reward. Annual Review of Psychology 57: 87-115. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/16318590
Scott, S. K., Young, A. W., Calder, A. J., Hellawell, D. J., Aggleton, J. P., & Johnson, M. (1997). Impaired auditory recognition of fear and anger following bilateral amygdala lesions. Nature 385: 254–257. http://www.cf.ac.uk/psych/home2/papers/aggleton/Nature%20-%20385%20-%20254257.pdf
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover, G. H., Kenna, H., et al. (2007). Dissociable intrinsic connectivity networks for salience processing and executive control. Journal of Neuroscience 27: 2349. http://neuro.cjb.net/cgi/content/abstract/27/9/2349
Siegel, A., Edinger, H., & Dotto, M. (1975). Effects of electrical stimulation of the lateral aspect of the prefrontal cortex upon attack behavior in cats. Brain Research 93: 473-484. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6SYR-4840TTX32G&_user=10&_coverDate=08%2F15%2F1975&_rdoc=1&_fmt=high&_orig=search& _origin=search&_sort=d&_docanchor=&view=c&_searchStrId=1509842536&_rerunOri gin=scholar.google&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md 5=847c9d2d236f8c1c60a7f4b0674543ee&searchtype=a
Siegel, A., Edinger, H., & Lowenthal, H. (1974). Effects of electrical stimulation of the medial aspect of the prefrontal cortex upon attack behavior in cats. Brain Research 66: 467-479. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6SYR-484DBF6D8&_user=10&_coverDate=02%2F08%2F1974&_rdoc=1&_fmt=high&_orig=search&_ origin=search&_sort=d&_docanchor=&view=c&_searchStrId=1509841573&_rerunOrigi n=scholar.google&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5= 35b6ce6d6686d719eef9f30bb51da726&searchtype=a
Simmons, W.K., Hamann, S.B., Harenski, C.N., Hu, X.P., & Barsalou, L.W. (2008). fMRI evidence for word association and situated simulation in conceptual processing. Journal of Physiology – Paris 102: 106-119. http://psychology.emory.edu/cognition/barsalou/onlinepapers.html
Smith S.M., Fox, P.T., Miller, K.L., Glahn, D.C., Fox, P.M., Mackay, C.E., et al. (2009). Correspondence of the brain’s functional architecture during activation and rest. Proceedings of the National Academy of Sciences 106: 13040–13045. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed
Spinks, J. A., Blowers, G. H., & Shek, D. T. (1985). The role of the orienting response in the anticipation of information: A skin conductance response study. Psychophysiology 22: 385-394. http://www.ncbi.nlm.nih.gov/pubmed/4023149
Sprengelmeyer, R. (2007). The neurology of disgust. Brain 130: 1715-1717.
http://www.ncbi.nlm.nih.gov/pubmed/17550907
Sprengelmeyer, R., Young, A. W., Schroeder, U., Grossenbacher, P. G., Federlein, J., Büttner, T., et al. (1999). Knowing no fear. Proceedings of the Royal Society B: Biological Sciences 266: 2451. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1690477/
Sprengelmeyer, R., Young, A.W., Calder, A.J., Karnat, A., Lange, H., Homberg, V., Perrett, D.I., Rowland, D. (1996). Loss of disgust. Perception of faces and emotions in Huntington's disease. Brain 119: 1647-1665. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed?term=Sprengelmeyer%20et%20al.%201996
Sprengelmeyer R, Rausch M, Eysel UT, Przuntek H. (1998). Neural structures associated with recognition of facial expressions of basic emotions. Proceedings of the Royal Society: Biological Sciences 265:1927-1931. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/9821359
Spreng, R.N., & Mar, R.A. (in press). I remember you: A role for memory in social cognition and the functional neuroanatomy of their interaction. Brain Research.
Sutton, S. K., & Davidson, R. J. (1997). Prefrontal brain asymmetry: A biological substrate of the behavioral approach and inhibition systems. Psychological Science 8: 204–210. http://www.jstor.org/stable/40063179?cookieSet=1
Suvak, M. K., & Barrett, L. F. (2011). Considering PTSD from the perspective of brain processes: A psychological construction analysis. Journal of Traumatic Stress 24: 3-24. http://www.affective-science.org/pubs/2011/suvak-barrett-2011.pdf
Suzuki, A., Hoshino, T., Shigemasu, K., & Kawamura, M. (2006). Disgust-specific impairment of facial expression recognition in Parkinson's disease. Brain 129: 707. http://brain.oxfordjournals.org/cgi/content/abstract/129/3/707
Svoboda, E., McKinnon, M.C., Levine, B. (2006). The functional neuroanatomy of autobiographical memory: A meta-analysis. Neuropsychologia 44: 2189–2208. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/16806314
Tomkins, S. S. (1962), Affect Imagery Consciousness: The Positive Affects (Vol. 1). New York: Springer.
Tomkins, S. S. (1963), Affect Imagery Consciousness: The Negative Affects (Vol. 2). New York: Springer.
Tsakiris, M., Hesse, M. D., Boy, C., Haggard, P., & Fink, G. R. (2007). Neural Signatures of Body Ownership: A Sensory Network for Bodily Self-Consciousness. Cerebral Cortex
17: 2235-2244. http://cercor.oxfordjournals.org/cgi/content/full/17/10/2235
Tsuchiya, N., Moradi, F., Felsen, C., Yamazaki, M., & Adolphs, R. (2009). Intact rapid detection of fearful faces in the absence of the amygdala. Nature Neuroscience 12: 1224-1225. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2756300/
Urry, H. L., van Reekum, C. M., Johnstone, T., Kalin, N. H., Thurow, M. E., Schaefer, H. S., et al. (2006). Amygdala and ventromedial prefrontal cortex are inversely coupled during regulation of negative affect and predict the diurnal pattern of cortisol secretion among older adults. The Journal of Neuroscience 26: 4415-4425. http://neuro.cjb.net/cgi/content/abstract/26/16/4415
Van der Horst, V.G., Holstege, G., 1998. Sensory and motor components of reproductive behavior: pathways and plasticity. Behavior and Brain Research 92: 157–167. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed
Van Snellenberg, J.X. & Wager, T.D. (in press). Cognitive and motivational functions of the human prefrontal cortex. In E. Goldberg and D. Bougakov (Eds). Luria’s Legacy in the 21st Century. New York: Oxford. http://www.columbia.edu/cu/psychology/tor/Papers/luria_vans_wager_sub.pdf
Vazdarjanova, A., & McGaugh, J. L. (1998). Basolateral amygdala is not critical for cognitive memory of contextual fear conditioning. Proceedings of the National Academy of Sciences 95: 15003-15007. http://www.pnas.org/content/95/25/15003.abstract
Vigneau, M., Beaucousin, V., Hervé, P. Y., Duffau, H., Crivello, F., Houdé, O., et al. (2006). Meta-analyzing left hemisphere language areas: phonology, semantics, and sentence processing. NeuroImage 30: 1414-1432. http://www.ncbi.nlm.nih.gov/pubmed/16413796
Vincent, J. L., Snyder, A. Z., Fox, M. D., Shannon, B. J., Andrews, J. R., Raichle, M. E., et al. (2006). Coherent spontaneous activity identifies a hippocampal-parietal memory network. Journal of Neurophysiology 96: 3517-3531. http://jn.physiology.org/cgi/content/abstract/96/6/3517
Vogt, B. A., Nimchinsky, E. A., Vogt, L. J., & Hof, P. R. (1995). Human cingulate cortex: Surface features, flat maps and cytoarchitecture. The Journal of Comparative Neurology
359: 490-506. http://onlinelibrary.wiley.com/doi/10.1002/cne.903590310/abstract
Vogt, B.A., Berger, G.R., & Derbyshire, S.W. (2003). Structural and functional dichotomy of human midcingulate cortex. European Journal of Neuroscience 18: 3134-3144. http://www.ncbi.nlm.nih.gov.ezp-prod1.hul.harvard.edu/pubmed/14656310
von dem Hagen, E.A., Beaver, J.D., Ewbank, M.P., Keane, J., Passamonti, L., Lawrence, A.D., Calder, A.J. (2009). Leaving a bad taste in your mouth but not in my insula. Social Cognitive Affective Neuroscience 4: 379-86. http://www.ncbi.nlm.nih.gov/pubmed/19505971
Vuilleumier, P. (2005). How brains beware: neural mechanisms of emotional attention. Trends in Cognitive Sciences 12: 585-594. http://www.ncbi.nlm.nih.gov.ezpprod1.hul.harvard.edu/pubmed/16289871
Vuilleumier, P., & Driver, J. (2007). Modulation of visual processing by attention and emotion: windows on causal interactions between human brain regions. Philosophical Transactions of the Royal Society B: Biological Sciences 362: 837. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2430001/
Vytal, K., & Hamann, S. (in press). Neuroimaging Support for Discrete Neural Correlates of Basic Emotions: A Voxel-based Meta-analysis. Journal of Cognitive Neuroscience.
Wager, T. D., & Smith, E. E. (2003). Neuroimaging studies of working memory: a metaanalysis. Cognitive, Affective & Behavioral Neuroscience 3: 255-274. http://www.columbiauniversity.org/cu/psychology/tor/Papers/Wager_Smith_2003_WM_ meta.pdf
Wager, T. D., Lindquist, M., & Kaplan, L. (2007). Meta-analysis of functional neuroimaging data: current and future directions. Social Cognitive and Affective Neuroscience 2: 150
158. http://scan.oxfordjournals.org/content/2/2/150.abstract
Wager, T. D., Phan, K. L., Liberzon, I., & Taylor, S. F. (2003). Valence, gender, and lateralization of functional brain anatomy in emotion: a meta-analysis of findings from neuroimaging. NeuroImage 19: 513-531. http://www.ncbi.nlm.nih.gov/pubmed/12880784
Wager, T. D., van Ast, V. A., Hughes, B. L., Davidson, M. L., Lindquist, M. A., & Ochsner, K. N. (2009a). Brain mediators of cardiovascular responses to social threat, Part II: Prefrontal-subcortical pathways and relationship with anxiety. Neuroimage 47: 836-851.
Wager, T. D., Waugh, C. E., Lindquist, M., Noll, D. C., Fredrickson, B. L., & Taylor, S. F. (2009b). Brain mediators of cardiovascular responses to social threat, Part I: Reciprocal dorsal and ventral sub-regions of the medial prefrontal cortex and heart-rate reactivity. Neuroimage 47: 821-835.
Wager, T. D., Reading, S., & Jonides, J. (2004). Neuroimaging studies of shifting attention: A meta-analysis. Neuroimage 22: 1679-1693. http://psych.colorado.edu/~tor/Papers/Wager_2004_Switching_Meta.pdf
Wager, T. D., Jonides, J., Smith, E. E., & Nichols, T. E. (2005). Towards a taxonomy of attention-shifting: Individual differences in fMRI during multiple shift types. Cognitive Affective and Behavioral Neuroscience 5: 127-143. http://psych.colorado.edu/~tor/Papers/Wager_2005_CABN_shifting_indivdiffs_fmri.pdf
Wager, T., Barrett, L.F., Bliss-Moreau, E., Lindquist, K.A., et al (2008). The neuroimaging of emotion. In M. Lewis, J.M. Haviland-Jones, & L.F. Barrett (Eds.), Handbook of emotions (3rd ed). New York: Guilford.
Wang, G., Tomasi, D., Backus, W., Wang, R., Telang, F., Geliebter, A., et al. (2008). Gastric distention activates satiety circuitry in the human brain. NeuroImage 39: 1824-1831. http://www.ncbi.nlm.nih.gov/pubmed/18155924
Warnick, J.E., LaPorte, J.L., Kalueff, A.V. (2010). Domain interplay in mice and men: new possibilities for the “Natural Kinds” theory of emotion. New Ideas in Psychology 29: 49
56.
Weierich, M. R., Wright, C. I., Negreira, A., Dickerson, B. C., & Barrett, L. F. (2010). Novelty as a dimension in the affective brain. NeuroImage 49:2871-2878. http://www.ncbi.nlm.nih.gov/pubmed/19796697
Whalen, P. J. (2007). The uncertainty of it all. Trends in Cognitive Sciences 11: 499–500.
http://www.whalenlab.info/Publications%20Page/PDFs/Whalen_TICS_2007.pdf
Whalen, P. J., Rauch, S. L., Etcoff, N. L., McInerney, S. C., Lee, M. B., & Jenike, M. A. (1998). Masked presentations of emotional facial expressions modulate amygdala activity without explicit knowledge. Journal of Neuroscience 18: 411. http://neuro.cjb.net/cgi/content/abstract/18/1/411
Whalen, P.J., Kagan, J., Cook, R.G., Davis, F.C., Kim, H., Polis, S., et al. (2004). Human amygdala responsivity to masked fearful eye whites. Science 306: 2061. http://www.whalenlab.info/Publications%20Page/PDFs/Whalen_etal_Sc_2004.pdf
Whalen, P.J., Shin, L.M., McInerney, S.C., Fischer, H., Wright, C.I., Rauch, S.L. (2001). A functional MRI study of human amygdala responses to facial expressions of fear vs. anger. Emotion 1: 70-83. http://www.whalenlab.info/Publications%20Page/PDFs/Whalen_Shin_2001.pdf
Wicker, B., Keysers, C., Plailly, J., Royet, J.P., Gallese, V, Rizzolati, G. (2003). Both of us disgusted in my insula: The common neural basis of seeing and feeling disgust. Neuron
40: 655–664. http://www.ncbi.nlm.nih.gov/pubmed/14642287
Wilson-Mendenhall, C.D., Barrett, L.F., Simmons, W.K., & Barsalou, L.W. (in press). Grounding emotion in situated conceptualization. Neuropsychologia.
http://psychology.emory.edu/cognition/barsalou/papers/Wilson_Mendenhall_et_alNeuropsychologia_in_press-grounding_emotion.pdf
Wilson, F. A. W., & Rolls, E. T. (1993). The effects of stimulus novelty and familiarity on neuronal activity in the amygdala of monkeys performing recognition memory tasks. Experimental Brain Research 93: 367–382.
http://www.springerlink.com/content/g222263510052042/
Wright, C. I., Fischer, H., Whalen, P. J., McInerney, S. C., Shin, L. M., & Rauch, S. L. (2001). Differential prefrontal cortex and amygdala habituation to repeatedly presented emotional stimuli. Neuroreport 12: 379-383.
http://journals.lww.com/neuroreport/Abstract/2001/02120/Differential_prefrontal_cortex _and_amygdala.39.aspx
Wright, C. I., Martis, B., Schwartz, C. E., Shin, L. M., Fischer H, H., McMullin, K., et al. (2003). Novelty responses and differential effects of order in the amygdala, substantia innominata, and inferior temporal cortex. NeuroImage 18: 660-669.
http://www.ncbi.nlm.nih.gov/pubmed/12667843
Wright, C. I., Negreira, A., Gold, A. L., Britton, J. C., Williams, D., & Barrett, L. F. (2008). Neural correlates of novelty and face-age effects in young and elderly adults. NeuroImage 42: 956-968. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2613685/
Wright, C. I., Wedig, M. M., Williams, D., Rauch, S. L., & Albert, M. S. (2006). Novel fearful faces activate the amygdala in healthy young and elderly adults. Neurobiology of Aging
27: 361-374. http://www.neurobiologyofaging.org/article/S0197-4580%2805%29000692/abstract
Wundt, W. (1998). Outlines of psychology (C. H. Judd, trans.). Bristol, UK: Thoemmes Press. (Original work published 1897).
Xu, F. (2002) The role of language in acquiring object kind concepts in infancy. Cognition 85: 223-250. http://babylab.berkeley.edu/ProfFeiXu.html
Yang, Y., & Raine, A. (2009). Neuroimaging: Prefrontal structural and functional brain imaging findings in antisocial, violent, and psychopathic individuals: A meta-analysis. Psychiatry Research 174: 81-88. http://www.ncbi.nlm.nih.gov/pubmed/19833485
Zahn, R., Moll, J., Paiva, M., Garrido, G., Krueger, F., Huey, E. D., et al. (2009). The neural basis of human social values: evidence from functional MRI. Cerebral Cortex 19: 276
283. http://cercor.oxfordjournals.org/cgi/content/abstract/19/2/276